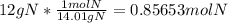Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 22:40
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization.a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution.part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1

Chemistry, 23.06.2019 02:20
Which of the following will cause an increase in the acceleration of an object? increase force decrease force increase mass decrease mass
Answers: 1

Chemistry, 23.06.2019 06:30
Aplanet similar to earth has four moons roughly the same distance away. the moon that will most affect tides on the planet is the one that has the greatest a) mass. b) volume. c) density. d) amount of water.
Answers: 1
You know the right answer?
How to convert 12g of nitrogen gas in mole...
Questions

Computers and Technology, 30.05.2020 00:02




Mathematics, 30.05.2020 00:02

Biology, 30.05.2020 00:02

Mathematics, 30.05.2020 00:02

Mathematics, 30.05.2020 00:02


Social Studies, 30.05.2020 00:02


Mathematics, 30.05.2020 00:02

Mathematics, 30.05.2020 00:02


Mathematics, 30.05.2020 00:02


Mathematics, 30.05.2020 00:02

Biology, 30.05.2020 00:02

Mathematics, 30.05.2020 00:02