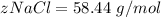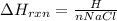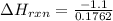Chemistry, 20.10.2020 20:01 Felici8617
A student dissolves 10,3 g of sodium chloride(NaCl) in 250 g of water in a well-insulated open cup. She then observes the temperature of the water fall from 23.0 °C to 22.2 °C over the course of 3 minutes. Use this data, and any information you need from the ALEKS Data resource,
NaCl (s) rightarrow Na+ (aq) + Cl- (aq)
You can make any reasonable assumptions about the physical properties of the solution. Note for advanced students: it's possible the student did not do the experiment carefully, and the values you calculate may not be the same as the known and published values for this reaction.
1. Is this reaction exothermic, endothermic, or neither?
2. If you said the reaction was exothermic or endothermic, calculate the amount of heat that was released or absorbed by the reaction in this case.
3. Calculate the reaction enthalpy delta Hrxn per mole of NaCl.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 14:00
Classify each statement about effective nuclear charge, zeff, as true or false.
Answers: 2

Chemistry, 22.06.2019 04:30
How many moles of air are there in a human lung with a volume of 2.4 l at stp? explain your answer
Answers: 1

Chemistry, 22.06.2019 05:50
According to coulomb's law, how would the electrical force between particles change if the product of their electrical charge increased?
Answers: 1

Chemistry, 22.06.2019 10:00
According to the tide table below what time of day will the highest tide occur? (2 pt) the highest tide will occur at
Answers: 1
You know the right answer?
A student dissolves 10,3 g of sodium chloride(NaCl) in 250 g of water in a well-insulated open cup....
Questions

Mathematics, 19.08.2020 19:01

Arts, 19.08.2020 19:01

World Languages, 19.08.2020 19:01

Mathematics, 19.08.2020 19:01



Mathematics, 19.08.2020 19:01


Mathematics, 19.08.2020 19:01

Mathematics, 19.08.2020 19:01

Mathematics, 19.08.2020 19:01



History, 19.08.2020 19:01


Computers and Technology, 19.08.2020 19:01

Geography, 19.08.2020 19:01

Mathematics, 19.08.2020 19:01

Mathematics, 19.08.2020 19:01

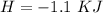
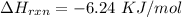

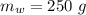
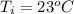


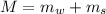
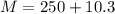
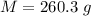
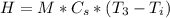
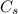 is the specific heat of solution which for this question we would assume it is the specific heat of water which has a value of
is the specific heat of solution which for this question we would assume it is the specific heat of water which has a value of 
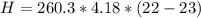

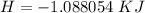

 is the molar mass of NaCl and the value is
is the molar mass of NaCl and the value is