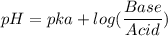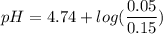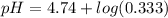Chemistry, 21.10.2020 16:01 anonymous115296
Calculate the expected pH of the buffer after the addition of 1.0 mL of 1M HCl. Remember that you are using 50 mL of the buffer, so be sure to calculate the moles of acetic acid and acetate in 50 mL of the buffer.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:30
18. use the activity series to predict whether the following synthesis reaction will occur. write the chemical equations for the reaction if it's predicted to occur. (s) + o2(g) -> *note: it is possible.*
Answers: 1

Chemistry, 22.06.2019 20:20
The characteristics of two different types of reactions are shown below: reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of an element. which statement is true about the atoms of the elements that participate in the two reactions? their identity changes in both reaction a and reaction b. their identity changes in reaction a but not in reaction b. their identity changes in reaction b but not in reaction a. their identity remains the same in both reaction a and reaction b.
Answers: 1


Chemistry, 23.06.2019 07:00
Write a hypothesis that answers the lesson question, “while observing a chemical reaction, how can you tell which reactant is limiting? ” hypothesis: if a substance is the limiting reactant, then . . because . .
Answers: 1
You know the right answer?
Calculate the expected pH of the buffer after the addition of 1.0 mL of 1M HCl. Remember that you ar...
Questions

Chemistry, 06.05.2020 07:26


Mathematics, 06.05.2020 07:26




History, 06.05.2020 07:26

Mathematics, 06.05.2020 07:27

Mathematics, 06.05.2020 07:27

History, 06.05.2020 07:27

English, 06.05.2020 07:27



Mathematics, 06.05.2020 07:27

Social Studies, 06.05.2020 07:27

Mathematics, 06.05.2020 07:27

Chemistry, 06.05.2020 07:27