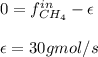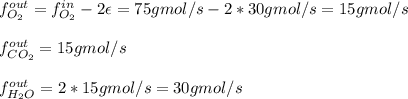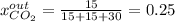Chemistry, 21.10.2020 16:01 HaileyAnn04
For the combustion of methane presented in Example 5.4, the chemical reaction is CH4 +2O2 →CO2 +2H2O Suppose that methane flows into a burner at 30 gmol/s, while oxygen flows into the same burner at 75 gmol/s. If all the meth- ane is burned and a single output stream leaves the burner, what is the mole fraction of CO2 in that output stream? Hint 1: Does the fact that all the methane is burned mean that all the oxygen is burned also? Hint 2: Find the molar flow rate of each component gas in the outlet gas ("flue gas").

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 20:30
In a laboratory experiment, a fermenting aqueous solution of glucose and yeast produces carbon dioxide gas and ethanol. the solution was heated by burning natural gas in a bunsen burner to distill the ethanol that formed in the flask. during the distillation, the ethanol evaporated and then condensed in the receiving flask. the flame of the burner was kept too close to the bottom of the flask and some of the glucose decomposed into a black carbon deposit on the inside of the flask. during this experiment the following changes occurred. which of these changes involved a physical change and not a chemical change? check all that apply. 1-condensation of ethanol 2-evaporation of ethanol 3- formation of carbon dioxide gas from glucose burning of natural gas 4-formation of ethanol from glucose by yeast 5-formation of a carbon deposit inside the flask
Answers: 2

Chemistry, 21.06.2019 23:00
Layers of rock containing fossils, like the layers illustrated here, are most likely composed of rocks.
Answers: 2


You know the right answer?
For the combustion of methane presented in Example 5.4, the chemical reaction is CH4 +2O2 →CO2 +2H2O...
Questions


English, 10.11.2020 02:20


English, 10.11.2020 02:20

Physics, 10.11.2020 02:20

Computers and Technology, 10.11.2020 02:20

Chemistry, 10.11.2020 02:20

Mathematics, 10.11.2020 02:20


Mathematics, 10.11.2020 02:20


Mathematics, 10.11.2020 02:20


Mathematics, 10.11.2020 02:20


Social Studies, 10.11.2020 02:20



Mathematics, 10.11.2020 02:20

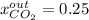
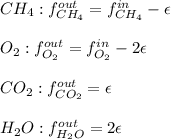
 accounts for the reaction extent. However, as all the methane is consumed, from the methane balance:
accounts for the reaction extent. However, as all the methane is consumed, from the methane balance: