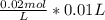Chemistry, 21.10.2020 17:01 cia196785920
Suppose a student needs to standardize a sodium thiosulfate, Na2S2O3,Na2S2O3, solution for a titration experiment. To do so, he or she will react it with a solution of iodine. The student adds a 1.00 mL1.00 mL aliquot of 0.0200 M KIO30.0200 M KIO3 solution to a flask, followed by 3 mL3 mL of distilled water, 0.2 g0.2 g of solid KI, KI, and 1 mL H2SO4.1 mL H2SO4. The student then titrates the solution with sodium thiosulfate solution in order to determine the exact concentration of Na2S2O3.Na2S2O3. The end point of the titration is reached after 0.90 mL0.90 mL of Na2S2O3Na2S2O3 is dispensed from a microburet. What is the concentration of the standard sodium thiosulfate solution?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 14:30
Alculate the concentration of h3o⁺in a solution that contains 5.5 × 10-5m oh⁻at 25°c. identify the solution as acidic, basic, or neutral.a) 1.8 × 10-10m, basicb) 1.8 × 10-10m, acidicc) 5.5 × 10-10m, neutrald) 9.2 × 10-1m, acidice) 9.2 × 10-1m, basic
Answers: 1


Chemistry, 22.06.2019 20:30
The activation energy for the reaction no2(g)+co2(g)⟶no(g)+co(g) is ea = 300 kj/mol and the change in enthalpy for the reaction is δh = -100 kj/mol . what is the activation energy for the reverse reaction?
Answers: 3

You know the right answer?
Suppose a student needs to standardize a sodium thiosulfate, Na2S2O3,Na2S2O3, solution for a titrati...
Questions

Mathematics, 30.08.2019 15:10


History, 30.08.2019 15:10

Mathematics, 30.08.2019 15:10



History, 30.08.2019 15:10





Mathematics, 30.08.2019 15:10

Physics, 30.08.2019 15:10


Mathematics, 30.08.2019 15:10

Chemistry, 30.08.2019 15:10