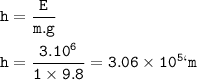Chemistry, 21.10.2020 21:01 bartam833oz4qy6
Burning 1 kg of coal releases about 3 million joules of energy. If you could use all of the chemical energy to lift another kilogram of coal, how high could you lift it?

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 12:00
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2

Chemistry, 22.06.2019 19:00
How does kepler second law of planetary motion overthrow one of the basic beliefs of classical astronomy
Answers: 1

Chemistry, 23.06.2019 01:00
Which description best characterization the motion of particles in a solid
Answers: 1
You know the right answer?
Burning 1 kg of coal releases about 3 million joules of energy. If you could use all of the chemical...
Questions






Mathematics, 02.11.2020 05:00



English, 02.11.2020 05:00

Arts, 02.11.2020 05:00


Mathematics, 02.11.2020 05:00

Physics, 02.11.2020 05:00






Mathematics, 02.11.2020 05:00

Mathematics, 02.11.2020 05:00