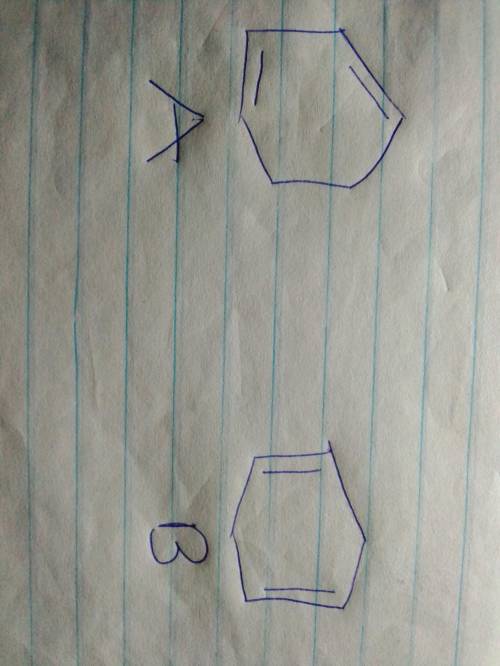PRACTICE PROBLEM Two compounds, A and B, have the same molecular formula, C6H8. Both A and B react with two molar equivalents of hydrogen in the presence of platinum to yield cyclohexane. Compound A shows three signals in its broadband decoupled NMR spectrum. Compound B shows only two NMR signals. Compound A shows an absorption maximum at 256 nm, whereas B shows no absorption maximum at wavelengths longer than 200 nm. What are the structures of A and B

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:00
If a polyatomic ionic compound has gained two hydrogen ions, then how does its name begin?
Answers: 3

Chemistry, 22.06.2019 18:00
How does climate change cause the ocean's thermohaline current to slow down?
Answers: 3

Chemistry, 22.06.2019 23:00
What is a substance? a. a physical property of matter b. a chemical property of matter c. an element or compound that cannot be physically separated d. characteristics used to tell the difference between mixtures
Answers: 1

Chemistry, 23.06.2019 00:40
To prevent the presence of air, noble gases are placed over highly reactive chemicals to act as inert "blanketing" gases. a chemical engineer places a mixture of noble gases consisting of 4.37 g of he, 13.36 g of ne, and 36.65 g of kr in a piston-cylinder assembly at stp. calculate the partial pressure in torr of kr.
Answers: 1
You know the right answer?
PRACTICE PROBLEM Two compounds, A and B, have the same molecular formula, C6H8. Both A and B react w...
Questions


English, 03.08.2019 11:00

Spanish, 03.08.2019 11:00


Mathematics, 03.08.2019 11:00

Mathematics, 03.08.2019 11:00


Computers and Technology, 03.08.2019 11:00

Health, 03.08.2019 11:00


Computers and Technology, 03.08.2019 11:00


History, 03.08.2019 11:00

History, 03.08.2019 11:00


History, 03.08.2019 11:00


Mathematics, 03.08.2019 11:00


Chemistry, 03.08.2019 11:00