Chemistry, 22.10.2020 19:01 pancakefox7
Suppose you are studying the kinetics of the iodine-catalyzed decomposition of hydrogen peroxide. 2 H 2 O 2 ⟶ 2 H 2 O + O 2 If you determine the initial rate is 7.50 × 10 − 4 M/s when [ H 2 O 2 ] = 0.546 M and [ K I ] = 0.212 M , what is the rate constant? Assume that the order of both reactants is 1.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 17:30
Write a paragraph at least 10 sentences explaining how rocks are used today in society
Answers: 1

Chemistry, 22.06.2019 04:00
14. many depressants reduce small muscle control, making it harder for a. you to steer b. your mind to consider complex problems c. the eye to scan, focus, or stay still d. the kidneys to filter alcohol out of the bloodstream
Answers: 3

Chemistry, 22.06.2019 13:00
What is the mass of 2.00 l of an intravenous glucose solution with a density of 1.15 g/ml?
Answers: 2

Chemistry, 22.06.2019 20:30
The activation energy for the reaction no2(g)+co2(g)⟶no(g)+co(g) is ea = 300 kj/mol and the change in enthalpy for the reaction is δh = -100 kj/mol . what is the activation energy for the reverse reaction?
Answers: 3
You know the right answer?
Suppose you are studying the kinetics of the iodine-catalyzed decomposition of hydrogen peroxide. 2...
Questions

History, 02.10.2021 23:20



Biology, 02.10.2021 23:20



Mathematics, 02.10.2021 23:20

Mathematics, 02.10.2021 23:20


Mathematics, 02.10.2021 23:20

History, 02.10.2021 23:20


Biology, 02.10.2021 23:20



Biology, 02.10.2021 23:20


Arts, 02.10.2021 23:20


Mathematics, 02.10.2021 23:20

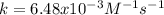
![r=k[H_2O_2][KI]](/tpl/images/0832/1312/1c29a.png)
![k=\frac{r}{[H_2O_2][KI]}=\frac{7.50x10^{-4}M/s}{0.546M*0.212M}\\ \\k=6.48x10^{-3}M^{-1}s^{-1}](/tpl/images/0832/1312/953ba.png)