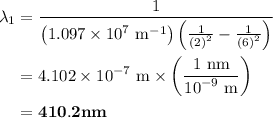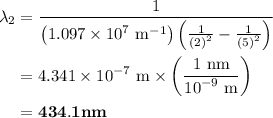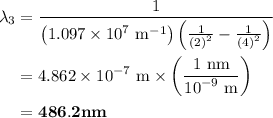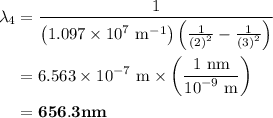Chemistry, 17.09.2019 12:20 lizzyhearts
The spectral lines observed for hydrogen arise from transitions from excited states back to the n=2 principle quantum level. calculate the wavelengths associated with the spectral transitions of the hydrogen atom from the n=6,5,4 and 3 to the n=2 level.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 12:00
In a laboratory, 1.55mg of an organic compound containing carbon, hydrogen, and oxygen is burned for analysis. this combustion resulted in the formation of 1.45mg of carbon dioxide and .89 mg of water. what is the empirical formula for this compound?
Answers: 1

Chemistry, 22.06.2019 14:00
Displacement is the slope of a velocity vs. time graph a. true b. false
Answers: 1

Chemistry, 22.06.2019 21:00
Use the measurements in the table to determine which unidentified metal has the highest density. metal volume mass a 10.5 cm3 122 g b 14.2 cm3 132 g c 16.1 cm3 115 g d 12.7 cm3 126 g
Answers: 2
You know the right answer?
The spectral lines observed for hydrogen arise from transitions from excited states back to the n=2...
Questions

Health, 30.10.2021 19:30

Mathematics, 30.10.2021 19:30

History, 30.10.2021 19:30

Biology, 30.10.2021 19:30

Mathematics, 30.10.2021 19:30

Mathematics, 30.10.2021 19:30

English, 30.10.2021 19:30


Mathematics, 30.10.2021 19:30

Mathematics, 30.10.2021 19:30


Social Studies, 30.10.2021 19:30

History, 30.10.2021 19:30

Mathematics, 30.10.2021 19:30

Mathematics, 30.10.2021 19:30



Biology, 30.10.2021 19:30


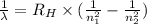
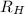 = Rydberg constant =
= Rydberg constant = 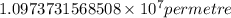
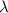 = wavelength
= wavelength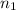 and
and 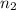 are the level of transitions.
are the level of transitions.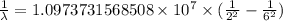
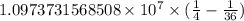
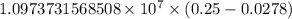
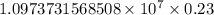
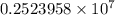
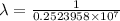


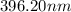
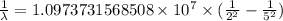
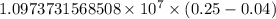
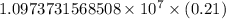
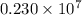
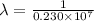


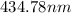
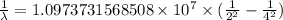
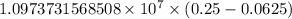
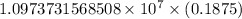
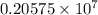
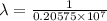


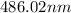
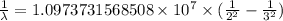
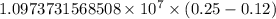
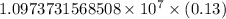
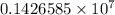
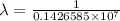


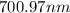
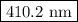 .
.
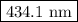 .
.
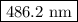 .
.
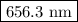 .
.
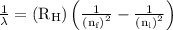 …… (1)
…… (1)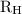 is the Rydberg constant that has the value
is the Rydberg constant that has the value 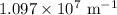 ,
, 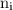 is the initial energy level of transition, and
is the initial energy level of transition, and 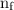 is the final energy level of transition.
is the final energy level of transition.
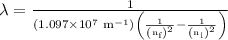 …… (2)
…… (2)