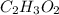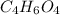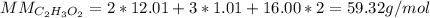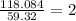Chemistry, 22.10.2020 22:01 tiffanyjadeb
Determine the empirical formula of a compound containing 40.6 grams of carbon, 5.1 grams of hydrogen, and 54.2 grams of oxygen.
In an experiment, the molar mass of the compound was determined to be 118.084 g/mol. What is the molecular formula of the compound?
For both questions, show your work or explain how you determined the formulas by giving specific values used in calculations. (10 points)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 15:00
20 pts ‼️ an unmanned spacecraft travels to mars. mars has a lower strength of gravity than earth. where in the image is the spacecraft’s weight the greatest?
Answers: 1

Chemistry, 22.06.2019 18:20
Which reason best explains why metals are malleable? a)because they have delocalized electrons b)because they have localized electrons c)because they have ionic bonds d)because they have rigid bonds
Answers: 2

Chemistry, 23.06.2019 06:10
How much would the freezing point of water decrease if 4 mol of nacl were added to 1 kg of water (kf=1.86 degrees c/(mol/kg) for water and i=2 for nacl a- 7.44 degrees c b- 14.88 c 3.72 d 1.86
Answers: 1

You know the right answer?
Determine the empirical formula of a compound containing 40.6 grams of carbon, 5.1 grams of hydrogen...
Questions

Health, 27.01.2020 17:31

History, 27.01.2020 17:31

History, 27.01.2020 17:31





Mathematics, 27.01.2020 17:31


Mathematics, 27.01.2020 17:31

History, 27.01.2020 17:31


Mathematics, 27.01.2020 17:31