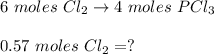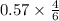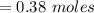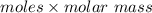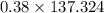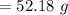Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:00
An alkaline battery produces electrical energy according to the following equation. zn(s) + 2 mno2(s) + h2o(l) zn(oh)2(s) + mn2o3(s) (a) determine the limiting reactant if 17.5 g zn and 31.0 g mno2 are used. (type your answer using the format ch4 for ch4.) (b) determine the mass of zn(oh)2 produced. _ g
Answers: 3


Chemistry, 22.06.2019 09:30
Right anwser gets marked brainliest newton's discovery concerning how fast an object will change speed is the: 1st law 2nd law 3rd law universal gravitation
Answers: 1

Chemistry, 22.06.2019 12:00
Ineed this asap part i: scientific method what is the difference between science and pseudoscience? what is the scientific method?
Answers: 2
You know the right answer?
A 12.39 g sample of phosphorus reacts with 40.75 g of chlorine to form only phosphorus trichloride (...
Questions


Mathematics, 13.11.2020 08:50

Computers and Technology, 13.11.2020 08:50

Advanced Placement (AP), 13.11.2020 08:50

Mathematics, 13.11.2020 08:50

History, 13.11.2020 08:50

History, 13.11.2020 08:50



Arts, 13.11.2020 08:50

Mathematics, 13.11.2020 08:50

English, 13.11.2020 08:50

Mathematics, 13.11.2020 08:50



Mathematics, 13.11.2020 08:50

Mathematics, 13.11.2020 08:50

Biology, 13.11.2020 08:50

Chemistry, 13.11.2020 09:00


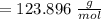
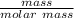
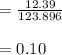
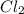 molar mass
molar mass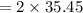
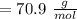
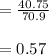
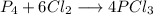
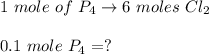
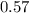 ,
, 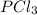 Theoretical performance
Theoretical performance