
Chemistry, 23.10.2020 19:20 Jazminruiz0527
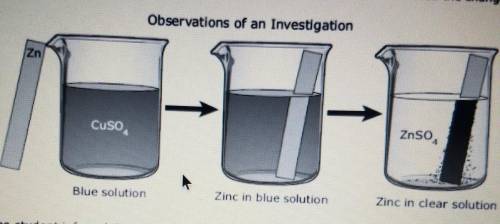
48 For an investigation a student poured a blue solution of Cuso, into a beaker. The student placed a shiny, silver-colored strip of zinc metal in the solution and observed the changes. Observations of an Investigation Zn Cuso, ZnSO4 Blue solution Zinc in blue solution Zinc in clear solution The student inferred that a chemical reaction occurred. What evidence supports this inference? F A dark solid formed on the zinc metal. G The zinc metal remained silver-colored and shiny. H The Cuso, solution turned blue when the zinc metal was added. None of these

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Asolution contains 225 g of sodium chloride, nacl, dissolved in enough water to make a 0.25 l of solution. what is the molarity of the solution?
Answers: 2

Chemistry, 22.06.2019 07:40
21. consider the following chemical reaction: n2+ o2 2 no if 10.0 g of n2 reacts with excess oxygen then how many grams of no can be formed? a) 10.7 g b) 21.4 g c) 32.9 g d) 42.8 g page 4 of 8
Answers: 2

Chemistry, 22.06.2019 16:10
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.300 m , [b] = 1.05 m , and [c] = 0.550 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.140 m and [c] = 0.710 m . calculate the value of the equilibrium constant, kc.
Answers: 1

You know the right answer?
48 For an investigation a student poured a blue solution of Cuso, into a beaker. The student placed...
Questions

Advanced Placement (AP), 13.10.2021 04:10

History, 13.10.2021 04:10

Biology, 13.10.2021 04:10


Mathematics, 13.10.2021 04:10

Mathematics, 13.10.2021 04:20




Chemistry, 13.10.2021 04:20


Mathematics, 13.10.2021 04:20

Advanced Placement (AP), 13.10.2021 04:20




Computers and Technology, 13.10.2021 04:20






