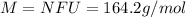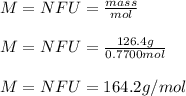Chemistry, 26.10.2020 16:40 melissapulido198
An 0.7700 mol sample of an ionic compound has a mass of 126.4 g. Determine the number of formula units of the compound that are present. Express answer in scientific notation.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:00
Schrodinger and heisenberg developed an alternate theory about atomic nature that contradicted some of bohr's model of the atom. how do changes resulting from new technology and evidence affect the reputation of the atomic theory?
Answers: 1


Chemistry, 22.06.2019 20:00
In vapor-liquid equilibrium in a binary mixture, both components are generally present in both phases. how many degrees of freedom are there for such a system? the reaction between nitrogen and hydrogen to form ammonia occurs in the gas phase. how many degrees of freedom are there for this system? steam and coal react at high temperatures to form hydrogen, carbon monoxide, carbon dioxide, and methane. the following reactions have been suggested as being involved in the chemical transformation:
Answers: 3

Chemistry, 23.06.2019 01:00
Which statement best describes isomers? a. isomers are alcohols that have the same functional group. b. isomers have at least one carbon-carbon double bond. c. isomers have the same molecular formula but different structural properties.
Answers: 1
You know the right answer?
An 0.7700 mol sample of an ionic compound has a mass of 126.4 g. Determine the number of formula uni...
Questions

Mathematics, 06.10.2019 04:30

Mathematics, 06.10.2019 04:30

Health, 06.10.2019 04:30








Mathematics, 06.10.2019 04:30

Mathematics, 06.10.2019 04:30






Spanish, 06.10.2019 04:30

Physics, 06.10.2019 04:30

English, 06.10.2019 04:30