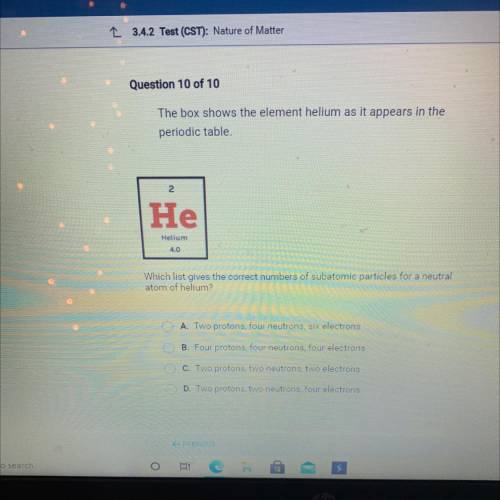
The box shows the element helium as it appears in the
periodic table
He
Helium
4....

The box shows the element helium as it appears in the
periodic table
He
Helium
4.0
Which list gives the correct numbers of subatomic particles for a neutral
atom of helium?
A. Two protons, four neutrons, six electrons
B. Four protons, four neutrons, four electrons
DC. Two protons, two
eutrons, two electrons
D. Two protons, two neutrons, four electrons

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:30
What effect might melting sea ice have for people who live in coastal areas?
Answers: 1

Chemistry, 22.06.2019 12:00
Solutions of sodium carbonate and silver nitrate react to form solid silver carbonate and a solution of sodium nitrate. a solution containing 3.50 g of sodium carbonate is mixed with one containing 5.00 g of silver nitrate. how many grams of sodium carbonate, silver nitrate, silver carbonate, and sodium nitrate are present after the reaction is complete?
Answers: 2

Chemistry, 22.06.2019 17:10
Calculate the estimated density of each ball. use the formula d = m/v where d is the density, m is the mass, and v is the volume. record your calculations in table a of your student guide. given that the density of water is 1.0 g/cm3, make a prediction about whether each ball will float in water. record your prediction in table a. what is the estimated density of the table tennis ball? record your answer to the nearest hundredth
Answers: 2

Chemistry, 22.06.2019 18:10
The atom fluorine generally will become stable through the formation of an ionic chemical compound by accepting electron(s) from another atom. this process will fill its outer energy level of electrons.
Answers: 1
You know the right answer?
Questions



English, 10.12.2021 19:00


English, 10.12.2021 19:00

Chemistry, 10.12.2021 19:00

Computers and Technology, 10.12.2021 19:00

Computers and Technology, 10.12.2021 19:00

History, 10.12.2021 19:00




Mathematics, 10.12.2021 19:00

Mathematics, 10.12.2021 19:00


History, 10.12.2021 19:00


Mathematics, 10.12.2021 19:00


Biology, 10.12.2021 19:00



