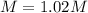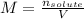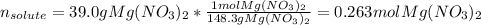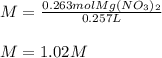Chemistry, 29.10.2020 17:00 angelcamacho0219
A chemist makes up a solution by dissolving 39.0 g of Mg(NO3)2 in enough water to produce a final solution volume of 257 mL. To calculate the molarity of the solution, what additional information is needed

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Explain why scientists use shared characteristics to make cladograms.
Answers: 1

Chemistry, 21.06.2019 23:40
If the atomic mass of an atom is 34 and the atom contains 13 protons, how many neutrons does the atom contain?
Answers: 2

Chemistry, 22.06.2019 01:00
Which statement correctly describes potassium iodide, ki? there is a one-to-one ratio of potassium ions to iodide ions. potassium gains electrons and iodine loses electrons during the reaction. the lattice is held together by potassium anions and iodide cations.
Answers: 1

Chemistry, 22.06.2019 20:30
Which states of matter have particles that move independently of one another with very little attraction?
Answers: 1
You know the right answer?
A chemist makes up a solution by dissolving 39.0 g of Mg(NO3)2 in enough water to produce a final so...
Questions


Mathematics, 01.12.2020 01:00



Computers and Technology, 01.12.2020 01:00

Mathematics, 01.12.2020 01:00

Chemistry, 01.12.2020 01:00




Health, 01.12.2020 01:00

Arts, 01.12.2020 01:00

Business, 01.12.2020 01:00

Mathematics, 01.12.2020 01:00



Mathematics, 01.12.2020 01:00

English, 01.12.2020 01:00