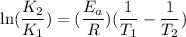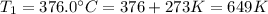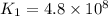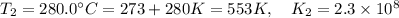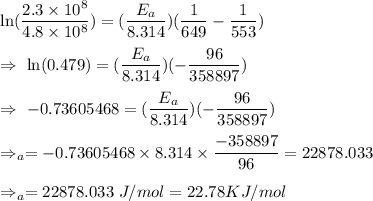The rate constant for a certain reaction is measured at two different temperatures:
Temperature K
376.0°C 4.8 x 10^8
280°C 2.3 x 10^8
Assuming the rate constant obeys the Arrhenius equation, calculate the activation energy for this reaction. Round your answer to significant digits.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:00
The most efficient way to establish the best possible economizer position is to measure
Answers: 1

Chemistry, 22.06.2019 05:30
Liv sheldon given the balanced equation for an organic reaction: c2h2 + 2cl2 → c2h2cl4 this reaction is best classified as *
Answers: 1


Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 2
You know the right answer?
The rate constant for a certain reaction is measured at two different temperatures:
Temperature K
Questions

Social Studies, 03.07.2019 10:50








Biology, 03.07.2019 10:50


Business, 03.07.2019 10:50




English, 03.07.2019 10:50


History, 03.07.2019 10:50

Biology, 03.07.2019 10:50


History, 03.07.2019 10:50

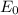 follows by formula:
follows by formula: