Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:00
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1

Chemistry, 22.06.2019 09:00
What term is missing from the central region that describes hypotheses, theories, and laws? popular predictable mathematical falsifiable
Answers: 2

Chemistry, 22.06.2019 16:10
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2

Chemistry, 22.06.2019 17:30
Oil rich countries in the middle east cover about 4% of earths total land area but prossess about 48% of the worlds known oil reserves what is the main reason for high concentration of reserves in this part of the world
Answers: 3
You know the right answer?
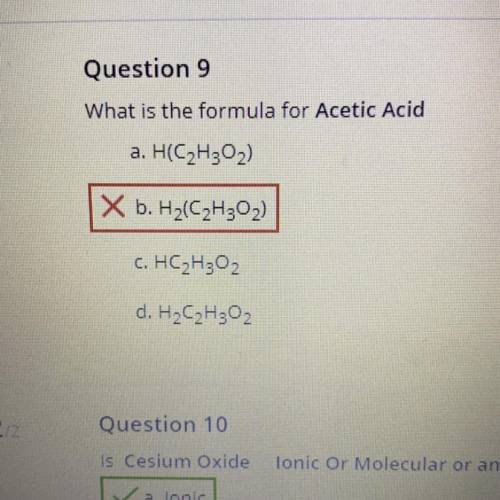
What is the formula for Acetic acid? (PLS USE ONE OF A, C, or, D) i have a test retake don’t let me...
Questions



Mathematics, 13.01.2021 22:40

History, 13.01.2021 22:40

History, 13.01.2021 22:40


Mathematics, 13.01.2021 22:40

History, 13.01.2021 22:40


History, 13.01.2021 22:40

Mathematics, 13.01.2021 22:40


History, 13.01.2021 22:40



Mathematics, 13.01.2021 22:40

Social Studies, 13.01.2021 22:40

English, 13.01.2021 22:40

Physics, 13.01.2021 22:40

Chemistry, 13.01.2021 22:40