
Chemistry, 29.10.2020 19:10 shanewugden7362
Theoretical element “A” has four isotopes. Use the data in the table to calculate the average atomic mass of the unknown element A. All steps of the calculations must be shown for credit.
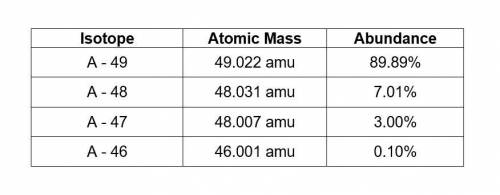

Answers: 1
Another question on Chemistry

Chemistry, 20.06.2019 18:04
1. according to the article, the chinese government has tried to control its people in all of the following ways except: question 1 options: create and spread propaganda in the media censorship of the media allowing people to create their own social media deleting people's social media posts without their permission question 2 (1 point) 2. when a government has total control over its citizens, including the media, it is a of government question 2 options: constitutional monarchy parliamentary democracy autocracy presidential democracy question 3 (1 point) according to the article, "social credit" is question 3 options: an experiment to people talk on-line a farming experiment that hurt people under mao ze dong using someone else's data from the internet, to give or take away things such as the right to buy an airline ticket a credit you get in china for having friends in real life, and not just on social media question 4 (1 point) china wants to take control over social media in which other region? question 4 options: taiwan chile japan hong kong
Answers: 1

Chemistry, 21.06.2019 20:00
Different isotopes indicate that an element will have different numbers of
Answers: 2

Chemistry, 22.06.2019 03:40
In an effort to address concerns about global warming, a power plant in portland,oregon is designed to take all of its exhaust gases from its boilers and recycle the co2 using the solvay process to make sodium hydrogen carbonate. the reaction is shown below. nh3(g) + h2o(l) + co2(g) + nacl(aq) → nahco3(aq) + nh4cl(aq) how many liters each of nh3 and co2 (both at stp) would be consumed to produce 3.00 kg of sodium bicarbonate? the volume of both nh3 and co2 would be
Answers: 1

You know the right answer?
Theoretical element “A” has four isotopes. Use the data in the table to calculate the average atomic...
Questions







Business, 28.11.2019 00:31















