
Chemistry, 30.10.2020 03:30 xxaurorabluexx
After two different elements are heated together the product has different chemical properties this is evidence that the two elements have
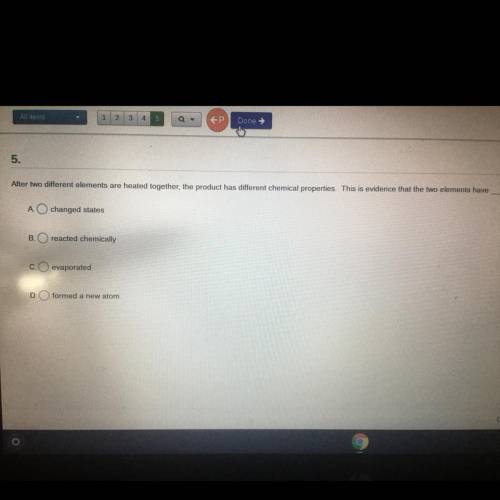

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 16:50
What is conserved in the reaction shown below? h2(g) + cl2 (g) --> 2hcl(g)a. mass onlyb. mass and moles onlyc. mass, moles, and molecules onlyd. mass, moles, molecules, and volume
Answers: 2

Chemistry, 22.06.2019 17:20
The small bags of silica gel you often see in a new shoe box are placed there to control humidity. despite its name, silica gel is a solid. it is a chemically inert, highly porous, amorphous form of sio2. because water vapor readily adsorbs onto the surface of silica gel, it acts as a desiccant. despite not knowing mechanistic details of the adsorption of water onto silica gel, from the information provided you should be able to make an educated guess about the thermodynamic characteristics of the process. predict the signs for δg, δh, and δs for the adsorption of water.
Answers: 2

Chemistry, 22.06.2019 21:00
Kp is the equilibrium constant for dissociation of the propionic acid dimer. what is the sign of the slope for a plot of the natural logarithm of kp vs. inverse temperature for this reaction?
Answers: 1

Chemistry, 23.06.2019 05:00
110 g of water (specific heat = 4.184 j/g c) and 100 g of a metal sample (specific heat = 0.397 j/g c) are heated from 25 degrees c to 75 degrees c. which substance required more thermal energy?
Answers: 1
You know the right answer?
After two different elements are heated together the product has different chemical properties this...
Questions

Mathematics, 24.06.2019 14:00




History, 24.06.2019 14:00




Mathematics, 24.06.2019 14:00

Mathematics, 24.06.2019 14:00

Social Studies, 24.06.2019 14:00




English, 24.06.2019 14:00

Mathematics, 24.06.2019 14:00



Biology, 24.06.2019 14:00

Biology, 24.06.2019 14:00



