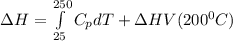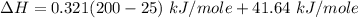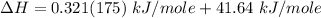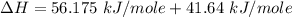Chemistry, 02.11.2020 16:30 monifaWilson
You are writing energy balances for a compound for which you cannot find heat capacity or latent heat data. All you know about the material are its molecular formula (C7H12N) and that it is a liquid at room temperature and has a normal boiling point of 200°C. Use this information to estimate the enthalpy of the vapor of this substance at 200°C relative to the liquid at 25°C.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 05:00
Use the table to identify the phase and phase changes of the elements under the given conditions. write the name of the substance, phase, or phase change
Answers: 3

Chemistry, 22.06.2019 08:00
Joe shines white light into a bowl half full of water at an angle of incident of 27.5°. calculate the angle of refraction in the water given the indices of refraction for air and water are 1.00 and 1.36, respectively.
Answers: 2

Chemistry, 22.06.2019 08:30
7. using your knowledge of colligative properties explain whether sodium chloride or calcium chloride would be a more effective substance to melt the ice on a slick sidewalk. use 3 – 4 sentences in your explanation. 8. when a 2.5 mol of sugar (c12h22o11) are added to a certain amount of water the boiling point is raised by 1 celsius degree. if 2.5 mol of aluminum nitrate is added to the same amount of water, by how much will the boiling point be changed? show all calculations leading to your answer or use 3 – 4 sentences to explain your answer. 9. if 5.40 kcal of heat is added to 1.00 kg of water at 100⁰c, how much steam at 100⁰c is produced? show all calculations leading to an answer. 10. the freezing of water at 0⁰c can be represented as follows: h2o (l) ↔ h2o(s) the density of liquid water is 1.00 g/cm3. the density of ice is 0.92 g/cm3. in 3 – 4 sentences explain why applying pressure causes ice to melt.
Answers: 1
You know the right answer?
You are writing energy balances for a compound for which you cannot find heat capacity or latent hea...
Questions


Advanced Placement (AP), 16.12.2020 18:30

Mathematics, 16.12.2020 18:30

Mathematics, 16.12.2020 18:30


Mathematics, 16.12.2020 18:30



Mathematics, 16.12.2020 18:30

Mathematics, 16.12.2020 18:30


Physics, 16.12.2020 18:30


Mathematics, 16.12.2020 18:30

Mathematics, 16.12.2020 18:30


World Languages, 16.12.2020 18:30



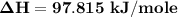
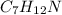
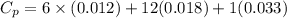
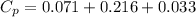
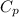 = 0.32 kJ/mole
= 0.32 kJ/mole