
Chemistry, 04.11.2020 02:30 Homepage10
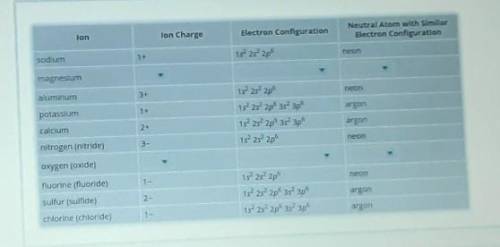
Recall that atoms gain or lose electrons to become ions. So, an ion has a different number of electrons than its corresponding neutral atom. Positive ions have lost electrons, and negative ions have gained electrons. The table includes the electron configuration of several ions. Complete the rows for magnesium and oxygen (oxide) ions by identifying the correct ion charge, electron configuration, and neutral atom with the same configuration as the ion. You may need to use online resources or the periodic table to determine the correct ion charges. Select the correct answer from each drop-down menu to complete the table.

Answers: 2
Another question on Chemistry



Chemistry, 22.06.2019 15:00
Describe what happens to the molecules as water goes from ice to liquid to vapor. be sure to explain what happens to the temperature during the phase changes.
Answers: 2

Chemistry, 22.06.2019 15:30
Which statement names the physical property of wood a. wood is softer than coal b. wood does not rust c. wood can rot d. wood can burn
Answers: 1
You know the right answer?
Recall that atoms gain or lose electrons to become ions. So, an ion has a different number of electr...
Questions

English, 22.04.2020 06:46


Spanish, 22.04.2020 06:46




Biology, 22.04.2020 06:47

Chemistry, 22.04.2020 06:47




Mathematics, 22.04.2020 06:47

Mathematics, 22.04.2020 06:48


English, 22.04.2020 06:48


Spanish, 22.04.2020 06:49


Social Studies, 22.04.2020 06:49



