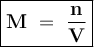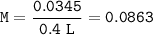Answers: 2
Another question on Chemistry



Chemistry, 21.06.2019 22:20
Asolution is made by dissolving 25.5 grams of glucose (c6h12o6) in 398 grams of water. what is the freezing point depression of the solvent if the freezing point constant is -1.86 °c/m? show all of the work needed to solve this problem.
Answers: 1

Chemistry, 22.06.2019 12:10
If a molecule with a molecular formula of c13h18 is treated with an excess of h2 in the presence of finally divided pt metal under conditions required for maximum hydrogenation of the molecule to give a molecule with a formula c13h24, how many rings are in the molecule?
Answers: 3
You know the right answer?
Cuanto es la molaridad de una solución que contiene 0.0345 moles NH4CL en exactamente 400.0 mL de so...
Questions



Mathematics, 09.09.2019 19:30


English, 09.09.2019 19:30


History, 09.09.2019 19:30


History, 09.09.2019 19:30




English, 09.09.2019 19:30



Mathematics, 09.09.2019 19:30


History, 09.09.2019 19:30


English, 09.09.2019 19:30