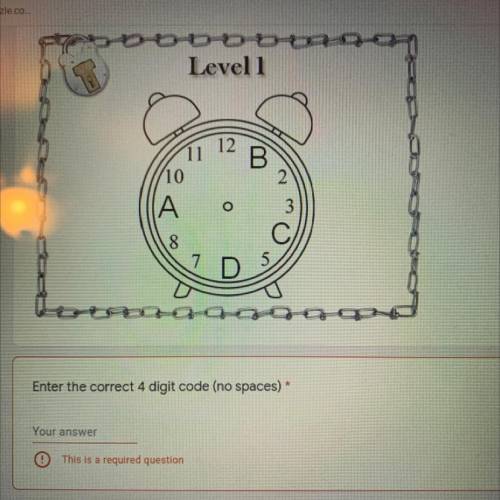
I need help asap please
...

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

Chemistry, 22.06.2019 13:30
Which statements are true concerning mineral formation? check all that apply. the slower the cooling, the larger the crystals. the faster the cooling, the smaller the crystals. crystals formed from magma are smaller than crystals formed from lava. minerals can only form in solutions when the solution is heated deep underground. when a solution cools, elements and compounds leave the solution and crystallize as minerals. minerals formed from hot water solutions can form narrow channels in the surrounding rock.
Answers: 1

Chemistry, 22.06.2019 16:30
How many grams of mgbr2 are needed to produce 75g or metal?
Answers: 1

Chemistry, 22.06.2019 17:30
To find the enthalpy of a reaction in the lab, you measured the of the reactants and the change during the reaction.
Answers: 1
You know the right answer?
Questions


Mathematics, 04.03.2021 01:00

English, 04.03.2021 01:00

History, 04.03.2021 01:00

History, 04.03.2021 01:00

Mathematics, 04.03.2021 01:00

Social Studies, 04.03.2021 01:00

Mathematics, 04.03.2021 01:00


Mathematics, 04.03.2021 01:00

Mathematics, 04.03.2021 01:00



Mathematics, 04.03.2021 01:00

Mathematics, 04.03.2021 01:00

Mathematics, 04.03.2021 01:00


Mathematics, 04.03.2021 01:00

Mathematics, 04.03.2021 01:00