
Chemistry, 04.11.2020 19:00 aredwolf2017
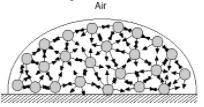
Surface tension is the inward pull that tends to minimize the surface area of a liquid. Water has high surface tension because of hydrogen bonding. In the diagram below, consider a molecule within the bulk of the liquid. This molecule experiences attractions to its neighboring molecules in all directions. These forces, shown by the arrows, average out to zero and there is no net force on the molecule. How is the situation different for a molecule at the surface? How does this give rise to surface tension?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 07:00
How many moles are in 7.2 x 10^23 carbon molecules? (*round to the nearest hundredth and include the unit "mol c" after your number) question 6 options:
Answers: 2


Chemistry, 22.06.2019 11:20
Sodium nitrite (nano2) reacted with 2−iodooctane to give a mixture of two constitutionally isomeric compounds of molecular formula c8h17no2 in a combined yield of 88%. draw reasonable structures for these two isomers. click the "draw structure" button to launch the drawing utility. place the two compounds in the appropriate boxes below.
Answers: 1

Chemistry, 22.06.2019 15:30
Two metal blocks that have slightly different temperatures are placed next to one another. after five minutes, they both have lower but equal temperatures. according to the law of conservation of energy, what most likelyhappened? energy was created inside the blocks.energy was destroyed inside the blocks.energy was absorbed into the blocks from outside the system.energy was transferred from the warmer block to the cooler block.
Answers: 2
You know the right answer?
Surface tension is the inward pull that tends to minimize the surface area of a liquid. Water has hi...
Questions






Mathematics, 11.02.2021 23:30




Mathematics, 11.02.2021 23:30

Spanish, 11.02.2021 23:30

Mathematics, 11.02.2021 23:30

English, 11.02.2021 23:30



History, 11.02.2021 23:30



Mathematics, 11.02.2021 23:30

History, 11.02.2021 23:30



