
Chemistry, 05.11.2020 16:40 MendesMist
Carbon monoxide and molecular oxygen react to form carbon dioxide. A 50.0 L reactor at 25.0 oC is charged with 1.00 bar of CO. The gas is then pressurized with O2 to give a total pressure of 3.52 bar. The reactor is sealed, heated to 350 oC to drive the reaction to completion, and cooled back to 25.0 oC. Compute the final partial pressure of each gas (in bar).

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:00
Select the correct answer. given: 2libr + ba → babr2 + 2li in this chemical reaction, 325 grams of barium (ba) react completely. how many moles of lithium (li) are produced? a. 1.18 mol b. 2.37 mol c. 4.73 mol d. 16.4 mol e. 32.9 mol
Answers: 2

Chemistry, 22.06.2019 08:40
Write the formula for the following chemicals. 7. e. trinitrogen tetraoxide a calcium phosphate f. magnesium acetate b. potassium sulfide g nickel(iii) cyanide c carbon dioxide h. silver sulfate d. cobalt(ii) chloride
Answers: 1

Chemistry, 22.06.2019 10:10
Stage in which a typical star has completely stopped fusion
Answers: 1

Chemistry, 22.06.2019 23:30
To find the work done, the force exerted and distance moved are multiplied. a couch is moved twice before you are happy with its placement. the same force was used to move the couch both times. if more work is done the first time it is moved, what do you know about the distance it was moved? a) when more work was done, the couch was moved the same distance. b) when more work was done, the couch was moved less. c) when more work was done, the couch was moved further. d) when more work was done, the couch wasn't moved at all.
Answers: 1
You know the right answer?
Carbon monoxide and molecular oxygen react to form carbon dioxide. A 50.0 L reactor at 25.0 oC is ch...
Questions

Chemistry, 22.04.2020 10:16

Physics, 22.04.2020 10:16

Health, 22.04.2020 10:16

Physics, 22.04.2020 10:16


Mathematics, 22.04.2020 10:16



Mathematics, 22.04.2020 10:17

Mathematics, 22.04.2020 10:17



Mathematics, 22.04.2020 10:17

English, 22.04.2020 10:17

Mathematics, 22.04.2020 10:17



English, 22.04.2020 10:17


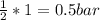 (required)
(required)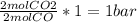 of CO2 2.52 bar O2 (initially) - 0.5 bar (reacted) = 2.02bar O2
of CO2 2.52 bar O2 (initially) - 0.5 bar (reacted) = 2.02bar O2

