
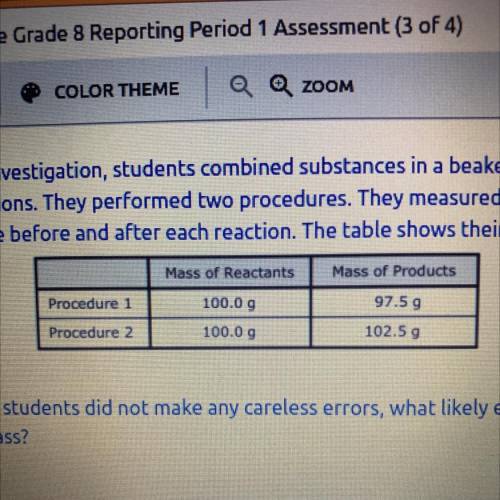
As part of an investigation, students combined substances in a beaker to observe
chemical reactions. They performed two procedures. They measured the mass of
each substance before and after each reaction. The table shows their observations.
Mass of Products
Procedure
97.59
Procedure 2
102.50
Procedure 1: All the reactants were liquids that evaporated.
Procedure 2: A gas was formed as one product, and it escaped into
the air
Mass of Reactants
100.0 9
100.00
Procedure 1: One of the reactants was converted to thermal
energy,
Procedure 2: All the products were liquids.
e
Assuming the students did not make any careless errors, what likely explains these
changes in mass?
Procedure 1: The reactants were liquids with different densities.
Procedure 2: The reactants were combined into only one product.
e
air.
Procedure 1: One of the products was a gas that escaped into the
Procedure 2: A gas from the air reacted with one of the other
reactants and formed a precipitate.
CLEAR ALL

Answers: 1
Another question on Chemistry



Chemistry, 22.06.2019 20:30
Some familiar products contain some of the same types of atoms. for instance, the chemical formula for baking soda is nahco 3. the chemical formula for liquid bleach is naclo, and the chemical formula for table salt is nacl. which choice best describes why these three products have some of the same types of atoms in common?
Answers: 1

You know the right answer?
As part of an investigation, students combined substances in a beaker to observe
chemical reactions...
Questions

English, 28.11.2020 08:10

Mathematics, 28.11.2020 08:10

English, 28.11.2020 08:10


Mathematics, 28.11.2020 08:10

Mathematics, 28.11.2020 08:10

Mathematics, 28.11.2020 08:10

Mathematics, 28.11.2020 08:10

Biology, 28.11.2020 08:10


Mathematics, 28.11.2020 08:10

History, 28.11.2020 08:10

Social Studies, 28.11.2020 08:10


Advanced Placement (AP), 28.11.2020 08:10

Health, 28.11.2020 08:10

Advanced Placement (AP), 28.11.2020 08:10

Mathematics, 28.11.2020 08:10

Biology, 28.11.2020 08:10

Biology, 28.11.2020 08:10



