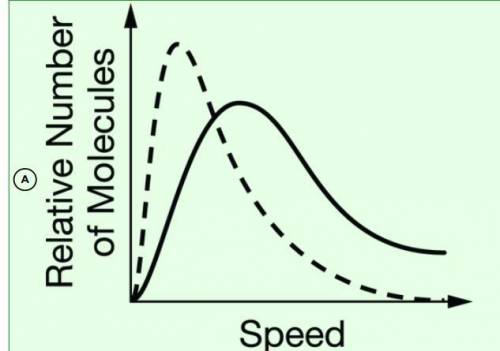
Chemistry, 06.11.2020 18:10 SpookyAlex2132
The diagram above shows the distribution of speeds for a sample of N2(g)
at 25°C
. Which of the following graphs shows the distribution of speeds for a sample of O2(g)
at 25°C
(dashed line) ?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:40
Kc = 0.040 for the system below at 450oc. if a reaction is initiated with 0.40 mole of cl2 and 0.40 mole of pcl3 in a 2.0 liter container, what is the equilibrium concentration of cl2 in the same system? pcl5(g) ⇄ pcl3(g) + cl2(g)
Answers: 3


Chemistry, 22.06.2019 09:40
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3

Chemistry, 22.06.2019 15:30
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 2
You know the right answer?
The diagram above shows the distribution of speeds for a sample of N2(g)
at 25°C
. Which of t...
. Which of t...
Questions

Mathematics, 12.07.2019 19:00



History, 12.07.2019 19:00

History, 12.07.2019 19:00

Mathematics, 12.07.2019 19:00




French, 12.07.2019 19:00

Social Studies, 12.07.2019 19:00


History, 12.07.2019 19:00

Biology, 12.07.2019 19:00




Mathematics, 12.07.2019 19:00

Social Studies, 12.07.2019 19:00