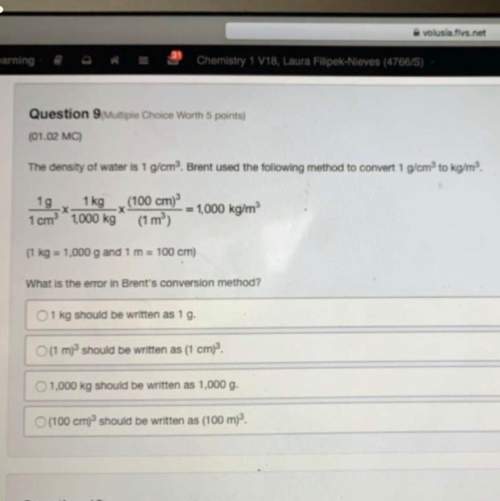
Chemistry, 06.11.2020 20:00 thibeauxkristy
A sample of 87.6 g of carbon is reacted with 136 g of
fluorine gas to produce carbon tetrafluoride. Using
the balanced equation below, predict which is the
limiting reactant and the maximum amount in moles
of carbon tetrafluoride that can be produced.
C +2F2 → CF4
A. fluorine, 1.79 moles
B. carbon, 7.29 moles
C. fluorine, 6.72 moles
D. carbon, 4.63 moles

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:30
Asample of ammonia reacts with oxygen as shown. 4nh3(g) + 5o2(g) 4no(g) + 6h2o(g) what is the limiting reactant if 4.0 g of nh3 react with 8.0 g of oxygen? o2 because it produces only 0.20 mol of no. nh3 because it produces only 0.20 mol of no. o2 because it produces two times less no than nh3. nh3 because it produces three times more no than o2.
Answers: 3

Chemistry, 22.06.2019 18:10
The atom fluorine generally will become stable through the formation of an ionic chemical compound by accepting electron(s) from another atom. this process will fill its outer energy level of electrons.
Answers: 1

Chemistry, 22.06.2019 19:00
Suppose that a certain fortunate person has a net worth of $71.0 billion ($7.10×1010). if her stock has a good year and gains $3.20 billion (3.20×109) in value, what is her new net worth?
Answers: 3

Chemistry, 23.06.2019 00:20
Which diagram represents the phase tha occurs after a solid melts?
Answers: 1
You know the right answer?
A sample of 87.6 g of carbon is reacted with 136 g of
fluorine gas to produce carbon tetrafluoride....
Questions





History, 17.01.2020 08:31


Mathematics, 17.01.2020 08:31

Mathematics, 17.01.2020 08:31


Mathematics, 17.01.2020 08:31



Mathematics, 17.01.2020 08:31

Computers and Technology, 17.01.2020 08:31

Mathematics, 17.01.2020 08:31

Mathematics, 17.01.2020 08:31


Mathematics, 17.01.2020 08:31


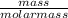
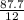 = 7.31moles
= 7.31moles 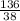 = 3.58moles
= 3.58moles 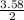 = 1.79moles of CF₄
= 1.79moles of CF₄