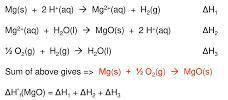Chemistry, 11.11.2020 17:50 skylar7192
What does Hess's law state can be done in order to be able to react solid magnesium with oxygen gas safely (that is, without exposing the magnesium to flames)?A. More moles of solid magnesium can be added until the reaction is safe. B. More moles of oxygen gas can be added until the reaction is safe. C. The reaction can be broken down and performed in steps. D. The phase state of the reactants can be changed to match each other. E. The phase state of the products can be changed to match each other.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:00
One does not belong why? ice, gold ,wood ,diamond and table salt
Answers: 1

Chemistry, 22.06.2019 14:00
The content of manganese (mn) in steel was determined spectrophotometrically and with the use of the standard addition method. an unknown sample of mn from a digested steel sample gave an absorbance of 0.185 when analyzed spectrophotometrically. when 5.00 ml of solution containing 95.5 ppm mn was added to 50.0 ml of the unknown steel solution (digested sample), the absorbance was 0.248. calculate the concentration, in parts-per-million (ppm), of mn in the digested steel sample solution.
Answers: 3

Chemistry, 22.06.2019 22:30
What relationship exists between an enzyme and a catalyst?
Answers: 1

Chemistry, 22.06.2019 22:30
3.09 lab: reaction of metals 1 which combinations of substances resulted in a chemical change? for each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. make a general statement about the reactivity of the metals in this experiment.
Answers: 1
You know the right answer?
What does Hess's law state can be done in order to be able to react solid magnesium with oxygen gas...
Questions


History, 26.08.2019 17:00


Health, 26.08.2019 17:00

Mathematics, 26.08.2019 17:00

History, 26.08.2019 17:00



History, 26.08.2019 17:00

History, 26.08.2019 17:00


Mathematics, 26.08.2019 17:00

Social Studies, 26.08.2019 17:00

History, 26.08.2019 17:00



Social Studies, 26.08.2019 17:00

English, 26.08.2019 17:00

English, 26.08.2019 17:00