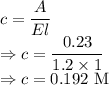Chemistry, 11.11.2020 17:50 raywils0n12300p0t3yc
A Beer's Law Plot of sample A gives a slope equal to 1.2 M-1cm-1. Calculate molar concentration of this sample at a wavelength of 420 nm and an absorbance reading of 0.23 in a 1 cm cuvette.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 14:00
Figure 10-1 study figure 10-1. the strong nuclear force felt by a single proton in a large nucleus
Answers: 3

Chemistry, 22.06.2019 09:10
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 17:10
Calculate the estimated density of each ball. use the formula d = m/v where d is the density, m is the mass, and v is the volume. record your calculations in table a of your student guide. given that the density of water is 1.0 g/cm3, make a prediction about whether each ball will float in water. record your prediction in table a. what is the estimated density of the table tennis ball? record your answer to the nearest hundredth
Answers: 2

Chemistry, 22.06.2019 23:00
Consider the reaction: 2al(s) + fe2o3(s) → al2o3(s) + 2fe(s) the δhf for fe2o3(s) = -824.3 kj/mole. the δhf for al2o3(s) = -1675.7 kj/mole. finish the equation. δhrxn = [(1)( kj/mole) + (2)( kj/mole)] - [(1)( kj/mole) + (2) ( kj/mole)]
Answers: 1
You know the right answer?
A Beer's Law Plot of sample A gives a slope equal to 1.2 M-1cm-1. Calculate molar concentration of t...
Questions

Geography, 30.03.2020 02:01



Mathematics, 30.03.2020 02:01

English, 30.03.2020 02:01


Biology, 30.03.2020 02:01



Mathematics, 30.03.2020 02:01

Mathematics, 30.03.2020 02:01




Mathematics, 30.03.2020 02:02

Mathematics, 30.03.2020 02:02


History, 30.03.2020 02:02

Mathematics, 30.03.2020 02:02

Mathematics, 30.03.2020 02:02

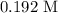
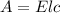
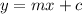
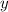 axis = A = Absorbance of solution = 0.23
axis = A = Absorbance of solution = 0.23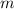 = Slope of line = El = Molar absorptivity multiplied with length =
= Slope of line = El = Molar absorptivity multiplied with length = 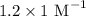
 = Value of x axis = c = Concentration of solution
= Value of x axis = c = Concentration of solution