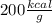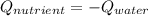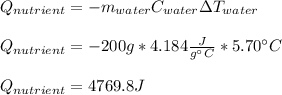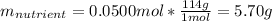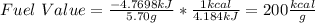Chemistry, 11.11.2020 23:20 khikhi1705
7.46 - A 0.0500-mol sample of a nutrient substance is burned in a bomb calorimeter containing 2.00 x 10g H20. If the formula weight of this nutrient substance is 114 g/mol, what is the fuel value (in nutritional Cal) if the temperature of the water increased 5.70C?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 07:00
What effect does a decrease in temperature have on the overall rate of a chemical reaction? a decrease in temperature decreases . the reaction rate will
Answers: 1

Chemistry, 22.06.2019 14:00
The content of manganese (mn) in steel was determined spectrophotometrically and with the use of the standard addition method. an unknown sample of mn from a digested steel sample gave an absorbance of 0.185 when analyzed spectrophotometrically. when 5.00 ml of solution containing 95.5 ppm mn was added to 50.0 ml of the unknown steel solution (digested sample), the absorbance was 0.248. calculate the concentration, in parts-per-million (ppm), of mn in the digested steel sample solution.
Answers: 3

Chemistry, 23.06.2019 00:00
What conclusion can you draw from this experiment about the components of the black ink?
Answers: 3

Chemistry, 23.06.2019 02:50
Dumbledore decides to gives a surprise demonstration. he starts with a hydrate of na2co3 which has a mass of 4.31 g before heating. after he heats it he finds the mass of the anhydrous compound is found to be 3.22 g. he asks everyone in class to determine the integer x in the hydrate: na2co3·xh2o; you should do this also. round your answer to the nearest integ
Answers: 2
You know the right answer?
7.46 - A 0.0500-mol sample of a nutrient substance is burned in a bomb calorimeter containing 2.00 x...
Questions

Advanced Placement (AP), 22.02.2021 22:10

Social Studies, 22.02.2021 22:10

Mathematics, 22.02.2021 22:10



Business, 22.02.2021 22:10

History, 22.02.2021 22:10

Mathematics, 22.02.2021 22:10

Mathematics, 22.02.2021 22:10



Mathematics, 22.02.2021 22:10


Mathematics, 22.02.2021 22:10

History, 22.02.2021 22:10

Mathematics, 22.02.2021 22:10

Spanish, 22.02.2021 22:10

English, 22.02.2021 22:10