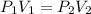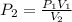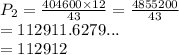Chemistry, 12.11.2020 22:30 thebasedgodchri
A gas at constant temperature has a pressure of 404.6 kPa with a volume of 12 ml. If the volume changes to 43ml, what is the new pressure

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 21:30
Put these processes of the water cycle in the correct order, starting at the point where the water is in the lake: 1. water evaporates into the atmosphere 2. rain, snow, or other precipitation falls 3. water collects into larger bodies of water 4. water vapor condenses into liquid water
Answers: 1

Chemistry, 22.06.2019 05:50
Fill in the coefficients that will balance the following reaction: a0cr2(so4)3 + a1agno3 -> a2cr(no3)3 + a3ag2so4
Answers: 1

Chemistry, 22.06.2019 07:20
Part b: study of equilibrium on solubility: mg(oh)2(s) ⇌ mg2+(aq) + 2 oh–(aq) cloudy clear (pink) 7. a. b. 8. a. b. 9. 10. 11. 12. when adding concentrated hydrochloric acid, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 7a. you should indicate which ion was added to or removed from the equilibrium mixture. when adding edta, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 8a. you should indicate which ion was added to or removed from the equilibrium mixture. upon heating in which direction is the equilibrium shifting? upon cooling in which direction is the equilibrium shifting? is the forward reaction a. endothermic explain your answers to questions 9, 10, and 11. (l) left (r) right (l) left (r) right b. exothermic
Answers: 1

Chemistry, 22.06.2019 08:30
Which of the following would have less momentum than a 52 kg cheetah running at 10 m/s?
Answers: 2
You know the right answer?
A gas at constant temperature has a pressure of 404.6 kPa with a volume of 12 ml. If the volume chan...
Questions


History, 17.07.2019 23:00


Chemistry, 17.07.2019 23:00

History, 17.07.2019 23:00


Biology, 17.07.2019 23:00


History, 17.07.2019 23:00

Biology, 17.07.2019 23:00


History, 17.07.2019 23:00


Social Studies, 17.07.2019 23:00


Business, 17.07.2019 23:00

Social Studies, 17.07.2019 23:00

Business, 17.07.2019 23:00

History, 17.07.2019 23:00