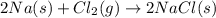Chemistry, 30.01.2020 05:01 dbn4everloved
Abalanced chemical equation indicates both the number of particles of reactants and products and the number of
a. orbits.
c. nuclei.
b. electrons.
d. moles

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 10:30
Great amounts of electromagnetic energy from our sun and other bodies in space travel through space. which is a logical conclusion about these electromagnetic waves? their energy must be very their frequency must be very low these waves can travel without a medium they only travel through a vacuum of space
Answers: 2

Chemistry, 22.06.2019 13:30
Which is true of a liquid? it has a definite volume but not a definite mass.it has a definite mass but not a definite volume.it has a definite volume but not a definite shape.it has a definite shape but not a definite volume.
Answers: 2

Chemistry, 22.06.2019 20:20
The characteristics of two different types of reactions are shown below: reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of an element. which statement is true about the atoms of the elements that participate in the two reactions? their identity changes in both reaction a and reaction b. their identity changes in reaction a but not in reaction b. their identity changes in reaction b but not in reaction a. their identity remains the same in both reaction a and reaction b.
Answers: 1

Chemistry, 22.06.2019 20:20
Which formula equation represents the burning of sulfur to produce sulfur dioxide? s(s) + o2(g) 4502(9) 2h2s(s) + 302(g) —> 2h20(0) + 2502(9) 4fes2+1102 —> 2fe2o3 + 8502 2802(g) + o2(9) v205 , 2503(9)
Answers: 1
You know the right answer?
Abalanced chemical equation indicates both the number of particles of reactants and products and the...
Questions

Mathematics, 31.03.2021 01:00

Mathematics, 31.03.2021 01:00

Chemistry, 31.03.2021 01:00

Mathematics, 31.03.2021 01:00


Mathematics, 31.03.2021 01:00

Mathematics, 31.03.2021 01:00

Mathematics, 31.03.2021 01:00


Mathematics, 31.03.2021 01:00


Mathematics, 31.03.2021 01:00

Mathematics, 31.03.2021 01:00

Chemistry, 31.03.2021 01:00

Mathematics, 31.03.2021 01:00


Mathematics, 31.03.2021 01:00

Mathematics, 31.03.2021 01:00