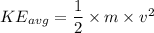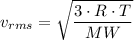The graph above shows the distribution of molecular speeds for four different gases at the same temperature. What property of the different gases can be correctly ranked using information from the graph, and why?
(Graph attached) I WILL MARK BRAINLIEST
A.) The densities of the gases, because as the density of a gas increases, the average speed of its molecules decreases.
B.) The pressures of the gases, because the pressure exerted by a gas depends on the average speed with which its molecules are moving.
C.) The volumes of the gases, because at a fixed temperature the volume of a gas can be calculated using the equation PV=nRT.
D.) The molecular masses of the gases, because the gas molecules have the same average kinetic energy and mass can be calculated using the equation KEavg=12mv2

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:00
What is the empirical formula of vanadium 1 oxide given that 20.38 grams of vandium combines with oxygen to form 23.58 grams of the oxide
Answers: 1

Chemistry, 22.06.2019 07:10
Remember to use the proper number of significant figures and leading zeros in all calculations.gelatin has a density of 1.27 g/cm³. if you have a blob of gelatin dessert that fills a 2.0 liter bottle, what is its mass? 2540 g2500 g3.9 x 10-43.937x 10-4
Answers: 3

Chemistry, 23.06.2019 06:30
What happens to the glucose molecule during the process of cellular respiration? (5 points) select one: a. it gets broken down. b. it forms oxygen. c. it builds muscles. d. it uses up energy.
Answers: 3

Chemistry, 23.06.2019 08:40
The half-life of a certain element is 100 days. how many half-lives will it be before only one eighth of this elementremains?
Answers: 1
You know the right answer?
The graph above shows the distribution of molecular speeds for four different gases at the same temp...
Questions

Mathematics, 04.03.2021 19:30

Mathematics, 04.03.2021 19:30



History, 04.03.2021 19:30


Mathematics, 04.03.2021 19:30

Chemistry, 04.03.2021 19:30


Mathematics, 04.03.2021 19:30

Mathematics, 04.03.2021 19:30


Computers and Technology, 04.03.2021 19:30


Mathematics, 04.03.2021 19:30


Advanced Placement (AP), 04.03.2021 19:30

Business, 04.03.2021 19:30

Mathematics, 04.03.2021 19:30

Mathematics, 04.03.2021 19:30