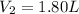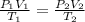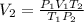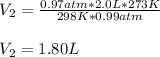A chemist conducts an experiment in which 2.0 L of hydrogen gas is collected over water at 1 atm and 298 K. Remember the pressure due to the hydrogen gas is 0.97 atm. Using the combined gas law, calculate the volume in L of the dry hydrogen gas at 273 K and a pressure of 1 atm.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 18:00
Construct the hypothetical phase diagram for metals a and b between room temperature (20c) and 700c, given the following information: * the melting temperature of metal a is 480c. • the maximum solubility of b in a is 4 wt% b, which occurs at 420c. • the solubility of b in a at room temperature is 0 wt% b. • one eutectic occurs at 420c and 18 wt% b–82 wt% a. • a second eutectic occurs at 475c and 42 wt% b–58 wt% a. • the intermetallic compound ab exists at a composition of 30 wt% b–70 wt% a, and melts congruently at 525c.• the melting temperature of metal b is 600c. • the maximum solubility of a in b is 13 wt% a, which occurs at 475c. • the solubility of a in b at room temperature is 3 wt% a.
Answers: 1

Chemistry, 22.06.2019 03:30
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 2

Chemistry, 22.06.2019 13:30
How many moles is 14.5 cm^3 of platinum? the density of platinum is 21.45 g/cm^3.
Answers: 1

You know the right answer?
A chemist conducts an experiment in which 2.0 L of hydrogen gas is collected over water at 1 atm and...
Questions


English, 12.11.2020 08:30



English, 12.11.2020 08:30

History, 12.11.2020 08:30

History, 12.11.2020 08:30


Mathematics, 12.11.2020 08:30


Physics, 12.11.2020 08:30

Social Studies, 12.11.2020 08:30

English, 12.11.2020 08:30

Mathematics, 12.11.2020 08:30


Mathematics, 12.11.2020 08:30

Health, 12.11.2020 08:30

History, 12.11.2020 08:30