
Chemistry, 18.11.2020 16:50 Conner4600
PLEASEEE HELPPP
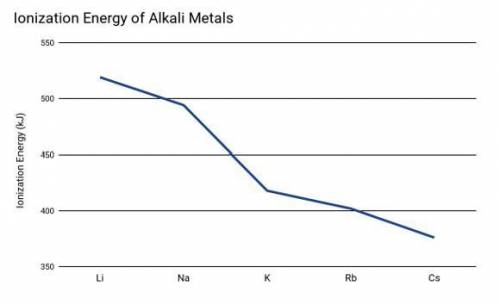
Two students were conducting and experiment based on the ionization energy of alkali metals. The driving question for the experiment was: "What is the relationship between ionization energy and the rate of reaction (time for the reaction to be completed) using alkali metals."
Which of the following predictions would be the best option based on the student's prior knowledge learned and information provided in the graph above?
A.
Cesium will have a higher rate of reaction (faster) because it has the lowest ionization energy which indicates a higher reactivity.
B.
Cesium will have a lower rate of reaction (slower) because it has the lowest ionization energy which indicates a lower reactivity.
C.
The ionization energy of the alkali metals will not affect the rate of the reaction because the energy required to remove an electron does not affect time.
D.
Lithium will have a higher rate of reaction (faster) because it has the highest ionization energy which indicates a higher reactivity.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 15:30
The wilson chamber is used to study: direction, speed, and distance of radioactivity the intensity of radiation the appearance of individual atoms all of the above
Answers: 1


Chemistry, 22.06.2019 04:00
Which of the following ocean acidification? are the most likely side effects of a ph less than 7.0 in the ocean b. more metal salts altering the ocean chemistry c. dissolution of the shells of marine organisms d. both a & b e. all of the above.
Answers: 3

Chemistry, 22.06.2019 04:50
Compare the equilibrium constants for the systems shown in the table. which favors products the most? which favors products the least? rank these systems in order from most to least in terms of favoring products rather than reactants. d > b > a > c c > a > b > d b > c > d > a a > d > c > b
Answers: 1
You know the right answer?
PLEASEEE HELPPP
Two students were conducting and experiment based on the ionization energy of alkal...
Questions


Mathematics, 14.07.2019 09:00



Computers and Technology, 14.07.2019 09:00


Mathematics, 14.07.2019 09:00







Mathematics, 14.07.2019 09:00

Biology, 14.07.2019 09:00

Mathematics, 14.07.2019 09:00

Health, 14.07.2019 09:00

English, 14.07.2019 09:00


Biology, 14.07.2019 09:00



