

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:10
According to the diagram; a) identify the anode of the cell and write the half-reaction that occurs there b) write the overall equation for the reaction that occurs as the cell operates c) calculate the value of the standard cell potential ,e cell. d)write the shorthand notation of the cell above e)indicate the flow of the electrons on the diagram
Answers: 3

Chemistry, 22.06.2019 07:30
Compare and contrast the bohr model and the electron cloud models of the atom.
Answers: 1

Chemistry, 22.06.2019 22:30
What is a number added in front of a formula in order to balance the equation
Answers: 1

Chemistry, 23.06.2019 10:20
Based on the equation, how many grams of br2 are required to react completely with 29.2 grams of alcl3? alcl3 + br2 → albr3 + cl2 48.7 grams 52.6 grams 56.7 grams 61.3 grams
Answers: 3
You know the right answer?
For the equilibrium that exists in an aqueous solution of nitrous acid (HNO2, a weak acid), the equi...
Questions







Computers and Technology, 09.07.2019 01:20





Mathematics, 09.07.2019 01:20

Arts, 09.07.2019 01:20



Health, 09.07.2019 01:20

Mathematics, 09.07.2019 01:20

Mathematics, 09.07.2019 01:20


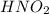
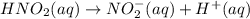
![k_a = \frac{[NO_{2}^{-}][H^{+}]}{HNO_{2}}](/tpl/images/0908/7640/67909.png)


