Look at the balanced equation below:
C2H6O + 3 O2 → 2 CO2 + 3 H2O
If you produced 11.5...


Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:50
In which situation can a mixture always be called a solution
Answers: 3

Chemistry, 22.06.2019 11:00
Which are examples of how technology has advanced scientific understanding.1using hot water to sterilize medical equipment.2transplanting a human organ into another individual.3inserting genes from one sheep into another cell to make a cloneunderstanding the different structures that make up a cell.4examining microorganisms from the deepest parts of the ocean
Answers: 2


Chemistry, 22.06.2019 23:00
Which organism develops breathing organism develops breathing organs from pharyngeal arches? shark, spider, sea star, sea horse
Answers: 2
You know the right answer?
Questions

Mathematics, 21.10.2020 01:01

English, 21.10.2020 01:01


Mathematics, 21.10.2020 01:01

Social Studies, 21.10.2020 01:01

Mathematics, 21.10.2020 01:01


History, 21.10.2020 01:01


Mathematics, 21.10.2020 01:01

Mathematics, 21.10.2020 01:01






Mathematics, 21.10.2020 01:01

Mathematics, 21.10.2020 01:01


Mathematics, 21.10.2020 01:01

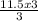 = 11.5moles of oxygen gas
= 11.5moles of oxygen gas

