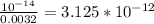Chemistry, 20.11.2020 14:00 sunshine52577oyeor9
Calculate the pH of the resulting solution if 32.0 mL of 0.320 M HCl(aq) is added to (a) 42.0 mL of 0.320 M NaOH(aq). (b) 22.0 mL of 0.420 M NaOH(aq).

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:50
Which of the following statements about acidic water is true? a. acid has no effect on the h,o molecules. b. the solution contains a larger number of oh ions than h,o ions. c. the solution contains a larger number of h,o ions than qh ions. d. the solution contains an equal number of h,o ions and oh ions. none of the above e.
Answers: 1

Chemistry, 22.06.2019 12:30
What is the percent composition of ca(oh)2? 37.7% ca, 53.0% o, and 10.3% h 45.5% ca, 38.2% o, and 16.3% h 54.0% ca, 43.0% o, and 2.7% h 64.7% ca, 27.0% o, and 8.3% h
Answers: 2


Chemistry, 22.06.2019 21:30
Harry lives in a city, and he has a lung condition known as asthma. on certain days, harry has to stay inside because pollutants in the air make it difficult for him to breathe. which of these pollution sources are nonpoint sources that might bother harry if he goes outside? choose the two that apply.
Answers: 3
You know the right answer?
Calculate the pH of the resulting solution if 32.0 mL of 0.320 M HCl(aq) is added to (a) 42.0 mL of...
Questions

Biology, 26.10.2020 17:40

History, 26.10.2020 17:40

Computers and Technology, 26.10.2020 17:40

Computers and Technology, 26.10.2020 17:40




Mathematics, 26.10.2020 17:40

Arts, 26.10.2020 17:40

Chemistry, 26.10.2020 17:40

Social Studies, 26.10.2020 17:40


Physics, 26.10.2020 17:40






Biology, 26.10.2020 17:40