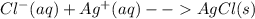Chemistry, 20.11.2020 16:30 jaidyn3mccoy6
A student analyzed an unknown sample that contained a single anion. The sample gave a white precipitate upon addition of a solution of AgNO3 which dissolved in ammonia. Which anion is present in the unknown solution? Write the corresponding net ionic equation.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:30
14. complete and balance the equations for the single displacement reactions. a. zn + pb(no3)2 -> b. al + niso4 -> 15. complete and balance the equations for the double displacement reactions. a. agno3(aq) + nacl(aq) -> b. mg(no3)2(aq) + koh(aq) -> 16. complete and balance the equations for the combustion reactions. a. __ ch4 + o2 -> b. __ c3h6 + o2 -> c. + o2 ->
Answers: 2

Chemistry, 21.06.2019 22:30
Which type of bond is present in hydrogen sulfide (h2s)? the table of electronegativities is given. a. hydrogen b. ionic c. nonpolar covalent d. polar covalent
Answers: 1

Chemistry, 22.06.2019 00:30
Used the balanced equation 2h2+ o2 - -> 2h2o. if you have 7.2 grams of o2 , how many grams of h2o can you produce ?
Answers: 2

Chemistry, 22.06.2019 04:50
The name of the ion, s2-, is: sulfurous ion sulfide ion sulfur ion sulfate ion
Answers: 1
You know the right answer?
A student analyzed an unknown sample that contained a single anion. The sample gave a white precipit...
Questions


Arts, 27.01.2021 19:40


Health, 27.01.2021 19:40





Mathematics, 27.01.2021 19:40



English, 27.01.2021 19:40

History, 27.01.2021 19:40

English, 27.01.2021 19:40

English, 27.01.2021 19:40

History, 27.01.2021 19:40


English, 27.01.2021 19:40


World Languages, 27.01.2021 19:40