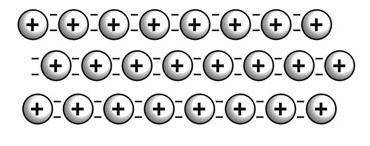
Which is true of the model of bonding shown in this figure?
Your
Metallic atoms are pr...

Chemistry, 20.11.2020 23:30 assassin42
Which is true of the model of bonding shown in this figure?
Your
Metallic atoms are present in a “sea” of negatively charged atoms.
Valence electrons are able to move easily among the metallic cations.
It results in the substance being very brittle and not easily deformed.
Protons are easily carried by the cations from one region to another.
Clear answer

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
1.aluminum chloride (alcl3), and sodium hydroxide (naoh) can react to form aluminum hydroxide (al(oh)3) and sodium chloride (nacl). you have 13.4 g of aluminum chloride and 10.0 g of sodium hydroxide. answer the following questions: •what is the balanced equation for this reaction? •if you use all 13.4 g of aluminum chloride, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •if you use all 10.0 g of sodium hydroxide, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •how many grams of aluminum hydroxide will actually be made? which reagent is limiting? explain your answer.
Answers: 1

Chemistry, 22.06.2019 05:40
Fill in the coefficients that will balance the following reaction: a0cr2(so4)3 + a1agno3
Answers: 3

Chemistry, 22.06.2019 12:30
The missing component to the table to the right or indicated with orange letters complete the table by filling in the corresponding numbers or symbols
Answers: 3

Chemistry, 22.06.2019 14:00
Will mark brainliest how many electrons can be held in the energy level n = 4?
Answers: 1
You know the right answer?
Questions



Mathematics, 13.10.2019 09:30

Mathematics, 13.10.2019 09:30



Mathematics, 13.10.2019 09:30


Mathematics, 13.10.2019 09:30

Biology, 13.10.2019 09:30


Mathematics, 13.10.2019 09:30

English, 13.10.2019 09:30

Biology, 13.10.2019 09:30

History, 13.10.2019 09:30

Mathematics, 13.10.2019 09:30

English, 13.10.2019 09:30


Computers and Technology, 13.10.2019 09:30

Geography, 13.10.2019 09:30



