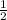Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:20
Which formula can be used to calculate the molar mass of ammonia (nh3)? molar mass of n + molar mass of h 3 × molar mass of n + molar mass of h molar mass of n + 3 × molar mass of h 3 × molar mass of n + 3 × molar mass of h
Answers: 1

Chemistry, 22.06.2019 06:00
Calculate the mass of silver needed to react with chlorine to produce 126g if silver chloride?
Answers: 3

Chemistry, 22.06.2019 09:00
Achemist 16 drop copper metal from copper chloride solution. the chemist place is 0.50 g of aluminum foil in a solution containing 0.75 g of copper (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction?
Answers: 1

Chemistry, 22.06.2019 09:40
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3
You know the right answer?
Write the balanced equation for the reaction that occurs when aluminum bromide is mixed with potassi...
Questions



Mathematics, 10.11.2020 22:10

Mathematics, 10.11.2020 22:10

Mathematics, 10.11.2020 22:10

History, 10.11.2020 22:10

Health, 10.11.2020 22:10


Mathematics, 10.11.2020 22:10

Social Studies, 10.11.2020 22:10


Mathematics, 10.11.2020 22:10



Mathematics, 10.11.2020 22:10

Mathematics, 10.11.2020 22:10

Social Studies, 10.11.2020 22:10


Social Studies, 10.11.2020 22:10