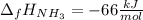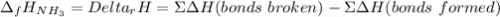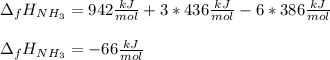Chemistry, 23.11.2020 02:30 afitzgerald
The formation of ammonia is represented by the equation N2(g) + 3H2(g) ⇌ 2NH3(g). Determine the enthalpy of formation of ammonia given the following mean bond enthalpies (kJmol-1): N≡N 942; H-H 436; N-H 386

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 21:30
Which statements are true about electrolysis? check all that apply. electrolysis requires an acid be present. electrolysis is described by two half-reactions. electrolysis is not an industrial process. electrolysis results in commercially valuable products. electrolysis involves the transfer of electrons. reduction results in the loss of electrons. oxidation results in the loss of electrons.
Answers: 1

Chemistry, 22.06.2019 10:10
How do you identify the anode on a power source such as a battery? how do you identify the cathode? how are terms anion and cation?
Answers: 1

Chemistry, 22.06.2019 12:00
A5.000 g sample of niso4 h2o decomposed to give 2.755 g of anhydrous niso4. what is the formula of the hydrate? what is the full chemical name for the hydrate? what is the molar mass of the hydrate? niso4•_h2o what is the mass % of water in the hydrate?
Answers: 1

Chemistry, 22.06.2019 23:30
With the largest atoms and the smallest number of valence electrons and with the smallest atoms and the greatest number of valence electrons are the most reactive. a. nonmetals; metals b. nonmetals; transition elements c. transition elements; metals d. metals; nonmetals
Answers: 3
You know the right answer?
The formation of ammonia is represented by the equation N2(g) + 3H2(g) ⇌ 2NH3(g). Determine the enth...
Questions

Mathematics, 24.04.2020 08:56

Mathematics, 24.04.2020 08:56



Mathematics, 24.04.2020 08:56


Biology, 24.04.2020 08:56

Mathematics, 24.04.2020 08:56

English, 24.04.2020 08:56


Physics, 24.04.2020 08:56




Mathematics, 24.04.2020 08:56



Mathematics, 24.04.2020 08:56

Chemistry, 24.04.2020 08:56