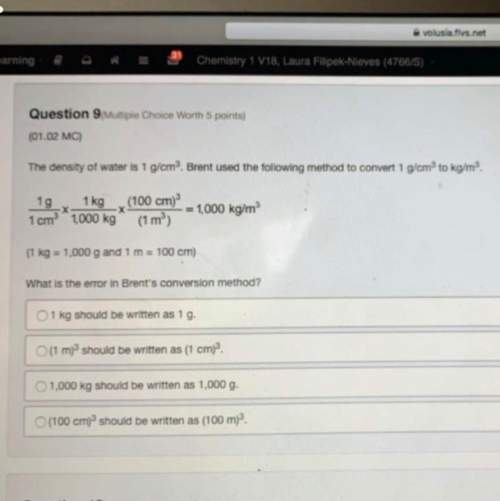
Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 12:50
Use the standard enthalpies of formation for the reactants and products to solve for the δhrxn for the following reaction. (the δhf of c2h4 is 52.26 kj/mol, co2 is -393.509 kj/mol, and h2o is -241.818 kj.) c2h4 (g) + 3o2(g) 2co2 (g) + 2h2o(g) δhrxn = the reaction is .
Answers: 3

Chemistry, 21.06.2019 20:00
Smog is the term used to describe the combination of fog and smoke
Answers: 1

Chemistry, 22.06.2019 08:30
Which metal exist in liquid state and can be cut with knife ?
Answers: 2

Chemistry, 22.06.2019 12:00
What is the percentage of hydrogen in nitrogen trihydride
Answers: 1
You know the right answer?
Write the balanced equation for the reaction that occurs when Iron (III) nitrate is mixed with magne...
Questions

History, 07.05.2021 21:40



Mathematics, 07.05.2021 21:40

Mathematics, 07.05.2021 21:40


Chemistry, 07.05.2021 21:40


Arts, 07.05.2021 21:40

Mathematics, 07.05.2021 21:40



Arts, 07.05.2021 21:40


English, 07.05.2021 21:40

Mathematics, 07.05.2021 21:40


Geography, 07.05.2021 21:40


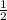 , d =
, d = 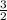 , b =
, b =