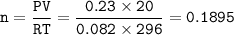Chemistry, 25.11.2020 14:00 cheyfaye4173
An Sulfur tetrafluoride gas is collected at 23.0 °C in an evacuated flask with a measured volume of 20.0 L. When all the gas has been collected, the pressure in the flask is measured to be 0.230 atm . Calculate the mass and number of moles of sulfur tetrafluoride gas that were collected. Be sure your answer has the correct number of significant digits. mass: 1 g N10 mole: mol X & ? Explanation Check

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:30
The table describes how some substances were formed substance 19 description formed by boiling pure water formed by combining three hydrogen atoms to every nitrogen atom formed by adding 5 g of sugar to 1 l of water formed by compressing carbon under high pressure based on the given descriptions, which substance is most likely a mixture?
Answers: 1

Chemistry, 22.06.2019 09:30
In apex! a liquid heated beyond a certain temperature becomes
Answers: 1

Chemistry, 22.06.2019 11:00
Which statement correctly identifies the scientific question and describes why the question is scientific? question 1 refers to the supernatural.question 2 reflects a moral or social value.question 3 refers to something that can be measured.question 4 reflects a question that can’t be observed.
Answers: 1

Chemistry, 22.06.2019 16:30
How many moles of sulfuric acid (h2so4) are needed to react completely with 6.8 moles of lithium hydroxide (lioh)? 2lioh + h2so4 → li2so4 + 2h2o a. 3.4 mol h2so4b. 6.8 mol h2so4 c. 10.2 mol h2so4 d. 13.6 mol h2so4
Answers: 3
You know the right answer?
An Sulfur tetrafluoride gas is collected at 23.0 °C in an evacuated flask with a measured volume of...
Questions

Mathematics, 19.05.2021 06:30


Mathematics, 19.05.2021 06:30

Biology, 19.05.2021 06:30





World Languages, 19.05.2021 06:30








History, 19.05.2021 06:30


English, 19.05.2021 06:30

Social Studies, 19.05.2021 06:30