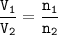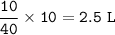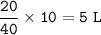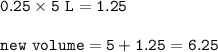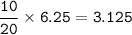The chemical equilibrium is given:
2NO (g) + O2(g) <—>2N O2, (g)
In a container...

Chemistry, 25.11.2020 14:00 shartman22
The chemical equilibrium is given:
2NO (g) + O2(g) <—>2N O2, (g)
In a container of volume 10L is in equilibrium a mixture consisting of 10 mol NO (g), 10 mol O2, (g) and 20 mol NO2, (g).
The volume of the container changes under constant temperature and after the restoration of equilibrium the amount of NO has increased by 25%. Calculate the volume change in L.
How do I find the change in volume?
P. s sorry if my English isn’t perfect.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 01:00
Which type of orbits are found in the principal energy level n = 2 a - s b - s, f c - s, d d - s, p e - s, p, d
Answers: 1

Chemistry, 22.06.2019 01:30
(apex) when a cup of water is dropped, as the cup falls, the water in the cup falls out true or false?
Answers: 1


Chemistry, 22.06.2019 10:30
Astudent reacts 13 moles of iron with 21 moles of oxygen according to the following equation:
Answers: 1
You know the right answer?
Questions

Chemistry, 29.01.2020 07:52

Geography, 29.01.2020 07:52


Mathematics, 29.01.2020 07:52





English, 29.01.2020 07:52

English, 29.01.2020 07:52

Mathematics, 29.01.2020 07:52

English, 29.01.2020 07:52

History, 29.01.2020 07:52

Biology, 29.01.2020 07:52



Mathematics, 29.01.2020 07:52