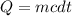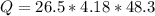Chemistry, 30.11.2020 07:00 TheJanko4526
1) How much energy, in joules, is needed to change 26.5 g of solid water at 0.0 deg C to liquid water at 48.3 deg C?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 13:30
Which statements are true concerning mineral formation? check all that apply. the slower the cooling, the larger the crystals. the faster the cooling, the smaller the crystals. crystals formed from magma are smaller than crystals formed from lava. minerals can only form in solutions when the solution is heated deep underground. when a solution cools, elements and compounds leave the solution and crystallize as minerals. minerals formed from hot water solutions can form narrow channels in the surrounding rock.
Answers: 1

Chemistry, 22.06.2019 21:30
How many liters of 3.0 m naoh solution will react with 0.60 liters of 4.0 m h2so4? h2so4 + naoh → na2so4 + h2o 1.2 l 1.6 l 2.4 l 2.8 l
Answers: 3

Chemistry, 23.06.2019 13:30
The activation energy for a(n) is quite large and usually takes extra energy from the environment, it is normally not a natural spontaneous process. combustion reaction endothermic reaction exothermic reaction catalyzed reaction
Answers: 1

Chemistry, 23.06.2019 15:50
Many radioactive atoms that have large masses undergo radioactive decay by releasing a particle that is identical to a helium-4 nucleus. what changes in the original atom are expected as a result of this natural phenomenon? the atomic number and the mass number will decrease. the atomic number and the mass number will increase. the atomic number will increase, and the mass number will decrease. the atomic number will decrease, and the mass number will increase.
Answers: 2
You know the right answer?
1) How much energy, in joules, is needed to change 26.5 g of solid water at 0.0 deg C to liquid wate...
Questions


Biology, 15.10.2020 14:01

Mathematics, 15.10.2020 14:01

English, 15.10.2020 14:01



Physics, 15.10.2020 14:01

Mathematics, 15.10.2020 14:01



English, 15.10.2020 14:01



English, 15.10.2020 14:01



Mathematics, 15.10.2020 14:01

Mathematics, 15.10.2020 14:01


Mathematics, 15.10.2020 14:01