Lab: Types of Chemical Reactions
Student Guide
This laboratory allows you to study various ki...

Lab: Types of Chemical Reactions
Student Guide
This laboratory allows you to study various kinds of chemical reactions, including some that result in precipitates.
Lesson Objectives
• Compare and contrast synthesis, single-displacement, and double-displacement reactions.
PREPARE
Approximate lesson time is 60 minutes.
Materials
• Lab Instructions: Lab_5.08_Instructions_modified_2020
• Lab Report: Lab_5.08_Report_modified_2020
• Lab Guidelines: Lab_Guidelines_modified
LEARN
Activity 1: Types of Chemical Reactions 1
Instructions
As you read through the lesson online, use the space below to take notes.
In this laboratory, you will study different kinds of chemical reactions.
Knowing the types of reactions helps you interpret your observations.
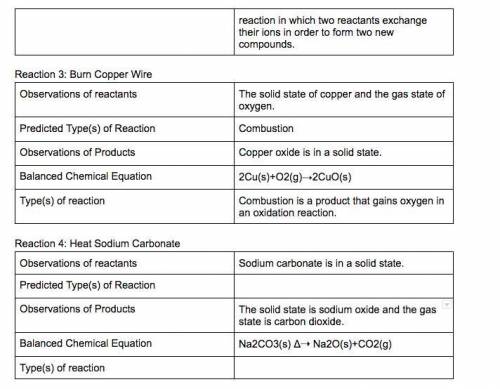
In a synthesis reaction, two reactants unite to form a third product.
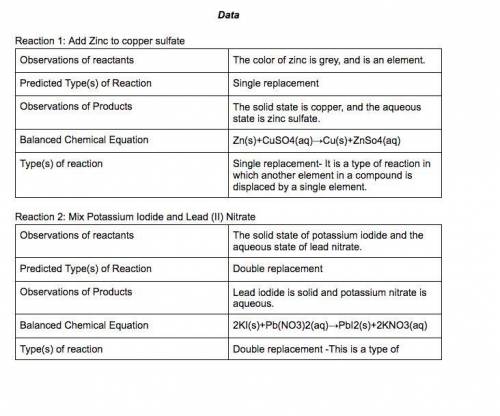
In a single-displacement reaction, one ion of a reactant bonds with the second reactant.
In a double-displacement reaction, ions of both reactants change places.
Activity 2: Types of Chemical Reactions 1
Instructions
Procedure
1. Open the Chemical Reactions Virtual Lab.
2. Click View the Tutorial and complete the tutorial to learn how to conduct the lab.
3. Close the tutorial and click begin the Lab.
Part 1 Synthesis Reaction
4. Perform the procedure, placing the magnesium strip in the flame.
5. Record your reaction.
6. Research the chemical reaction of magnesium and oxygen gas. Write an equation for the chemical reaction
that accounts for the observed reaction in this part of the lab.
7. Answer the question: What is a synthesis reaction?
8. Answer the questions on Part 1 in the Lab Report.
Part 2 Single Displacement Reaction
9. Place 1 scoop of zinc in Vial A and add 10 drops of copper (II) sulfate. Observe the reaction.
10. Place ball of aluminum in Vial B and add 10 drops of copper (II) sulfate. Observe the reaction.
11. Place 1 scoop of zinc in Vial C and add 10 drops of silver nitrate. Observe the reaction.
12. Place copper wire in Vial D and add 10 drops of silver nitrate, wait 5 minutes. Observe the reaction.
13. Complete the ta

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:30
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1

Chemistry, 22.06.2019 10:30
What woukd most likely be the transmittance at a 0.70 m solution of solute a? a) 7.6%b) 1.1%c)4.0%d)4.6%
Answers: 1

Chemistry, 22.06.2019 12:30
Consider the four elements above. which one of these elements will combine with oxygen in a 1: 1 ratio?
Answers: 3

Chemistry, 22.06.2019 17:30
A650 ml sodium bromine solution has a bromide ion concentration of 0.245 m. what is the mass (g) of sodium bromide in solution? a) 103.b)0.00155.c)16400.d) 16.4.e) 0.159
Answers: 2
You know the right answer?
Questions






English, 26.01.2021 16:10

Mathematics, 26.01.2021 16:10


Mathematics, 26.01.2021 16:10

English, 26.01.2021 16:10

English, 26.01.2021 16:10


Social Studies, 26.01.2021 16:10

Mathematics, 26.01.2021 16:10

Mathematics, 26.01.2021 16:10


Physics, 26.01.2021 16:10


Mathematics, 26.01.2021 16:10

Mathematics, 26.01.2021 16:10