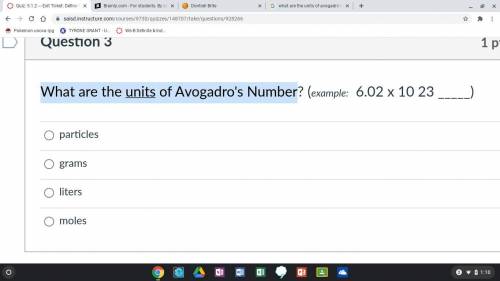
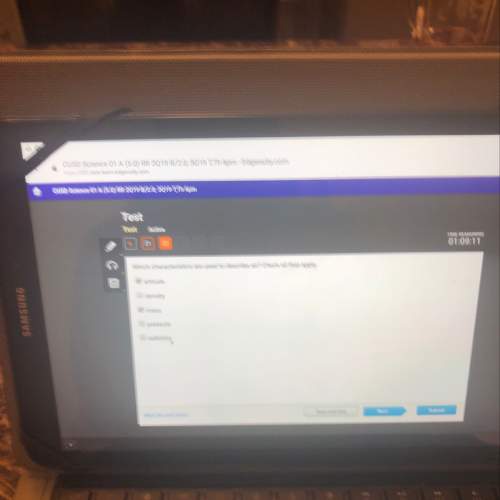
Which Answer Choice is right
...

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 22:30
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3

Chemistry, 23.06.2019 07:10
1) a light bulb takes in 30 of energy per second. it transfers 3j as use energy. calculate the efficiency. second. it transfers 3j as useful light energy and 27j as heat energy. calculate the efficiency
Answers: 1

Chemistry, 23.06.2019 08:30
Imagine you are a business executive who wants to pursue an environment policy for your company that limits pollution and uses fewer raw materials but would cost more what might be the discussion to your next broad meeting how would you make your case to your shareholders
Answers: 1
You know the right answer?
Questions

History, 16.10.2020 23:01

Mathematics, 16.10.2020 23:01

Mathematics, 16.10.2020 23:01


Chemistry, 16.10.2020 23:01



History, 16.10.2020 23:01


History, 16.10.2020 23:01



History, 16.10.2020 23:01


Mathematics, 16.10.2020 23:01

Mathematics, 16.10.2020 23:01

History, 16.10.2020 23:01



Mathematics, 16.10.2020 23:01