PLEASE HELP
give the electron configuration of vanadium (V), atomic number 23
Give the...

PLEASE HELP
give the electron configuration of vanadium (V), atomic number 23
Give the noble gas configuration of vanadium (V), atomic number 23
List the energy levels for the orbital configuration of vanadium (V), atomic number 23
How does the atomic radius change going down and across the periodic table?
How does the first ionization energy change going down and across the periodic table?
How does electronegativity change going down and across the periodic table?
How does the radius of a positive and negative ion compared to a neutral atom?
Match: A. Ionic bond B. Covalent bond C. Metallic bond __sharing of electrons __freely moving electrons __transfer electrons
Describe the dipole dipole force
Describe hydrogen bonding
Describe the Van der waals forces
Imagine you need to take a medicine that the doctor has prescribed for you. Explain why scientist who developed that medicine would need to know whether or not the compound in that medicine is polar. How might a polar medicine behave differently within your body than a non-polar medicine word? (Answer in 1 to 2 paragraphs)
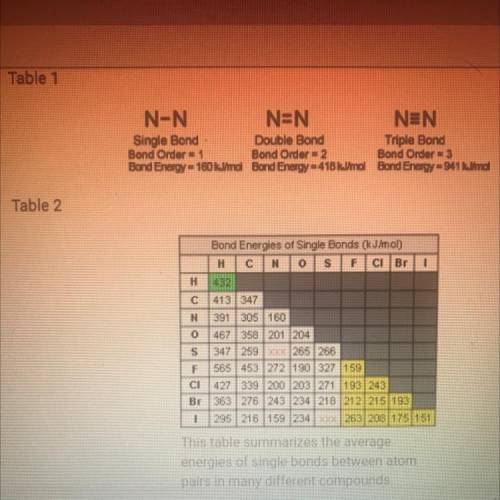
According to table 2, which is the strongest bond? Which is the week is fine? Based on what you know about the atomic radii and electronegativity of the elements involved in the bonds, why do you think these to have the most extreme bond-energy values?
How are the bond energies of each bond listed in table two determined?
Why do you think there aren’t bond energy values given in table 2 for N-S and S-I?
Based on tables one and two how would you describe the trend in bond strength of single double and triple bonds?
Based on table 2, how would you describe the trend in strength of bonds formed by the elements carbon nitrogen and oxygen? Would you describe this trend as a periodic trend? Why or why not?
What is the VSEPR theory?
How does electron repulsion determine molecular shape?
How do lone electron pairs affect molecular shape?
Draw the Lewis structure for the Se and 2 H atoms
Draw the Lewis structure for the SeH(2) molecule
What shape would SeH(2) have? Draw the molecule
Identify each of the following as a covalent compound, or ionic compound. Then provide either the formula for compounds identified by name or the name for those identified by formula:
Li(2)O
Dibitrogen trioxide
PCl(3)
Manganese(|||) oxide
Calcium bromide
THANK YOU FOR YOUR HELP <3

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:00
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible? a. attractive forces between gas particles are negligible because the particles of an ideal gas are moving so quickly. b. collisions between gas particles are elastic; there is no net gain or loss of kinetic energy. c. gases consist of a large number of small particles, with a lot of space between the particles. d. gas particles are in constant, random motion, and higher kinetic energy means faster movement.
Answers: 1

Chemistry, 22.06.2019 03:10
Agas diffuses 1/7 times faster than hydrogen gas (h2). what is the molar mass of the gas? 100.10 g/mol 98.78 g/mol 86.68 g/mol 79.98 g/mol
Answers: 3

Chemistry, 22.06.2019 09:30
Based on its chemical properties, identify the position of each chemical family on the periodic table.
Answers: 3

Chemistry, 22.06.2019 15:30
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 2
You know the right answer?
Questions

Mathematics, 23.06.2019 18:50


Computers and Technology, 23.06.2019 18:50




Mathematics, 23.06.2019 18:50


History, 23.06.2019 18:50

World Languages, 23.06.2019 18:50

Mathematics, 23.06.2019 18:50


History, 23.06.2019 18:50


Computers and Technology, 23.06.2019 18:50


Mathematics, 23.06.2019 18:50

Mathematics, 23.06.2019 18:50






